Type IIA - Type IIB protein tyrosine kinase inhibitors hybridization as an efficient approach for potent multikinase inhibitor development: Design, synthesis, anti-proliferative activity, multikinase inhibitory activity and molecular modeling of novel ...
Association between the saphenous vein diameter and venous reflux on computed tomography venography in patients with varicose veins | PLOS ONE
FPL - Turkey Point Units 6 & 7 COLA (Enclosures) - Part 11 Enclosure 8-Supplemental Field Investigation Data Report-Pages A-

Synthesis and Characterization of Organic Dyes with Various Electron‐Accepting Substituents for p‐Type Dye‐Sensitized Solar Cells - Weidelener - 2014 - Chemistry – An Asian Journal - Wiley Online Library

Suburethral Synthetic Adjustment-controlled Tape Compared with Conventional Treatment for Female Stress Urinary Incontinence: A Randomized Controlled Trial - European Urology Focus

Statistical Analyses of Incidence and Rates of Cardiovascular Disease... | Download Scientific Diagram
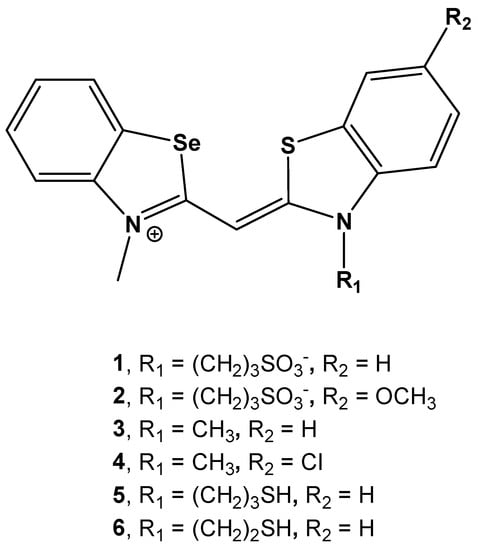
Biomolecules | Free Full-Text | Selenium-Substituted Monomethine Cyanine Dyes as Selective G-Quadruplex Spectroscopic Probes with Theranostic Potential

Amplification of local chirality within a folded dendrimer. An intramolecular 'sergeants and soldiers' experiment | Proceedings of the Royal Society A: Mathematical, Physical and Engineering Sciences

Type IIA - Type IIB protein tyrosine kinase inhibitors hybridization as an efficient approach for potent multikinase inhibitor development: Design, synthesis, anti-proliferative activity, multikinase inhibitory activity and molecular modeling of novel ...

Cyclometalation of 6-Phenyl-2,2′-Bipyridine and Iridium: Synthesis, Characterization, and Reactivity Studies | Organometallics
![Discovery of Thieno[2,3-d]pyrimidine-based KRAS G12D inhibitors as potential anticancer agents via combinatorial virtual screening - ScienceDirect Discovery of Thieno[2,3-d]pyrimidine-based KRAS G12D inhibitors as potential anticancer agents via combinatorial virtual screening - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0223523422001453-sc1.jpg)
Discovery of Thieno[2,3-d]pyrimidine-based KRAS G12D inhibitors as potential anticancer agents via combinatorial virtual screening - ScienceDirect
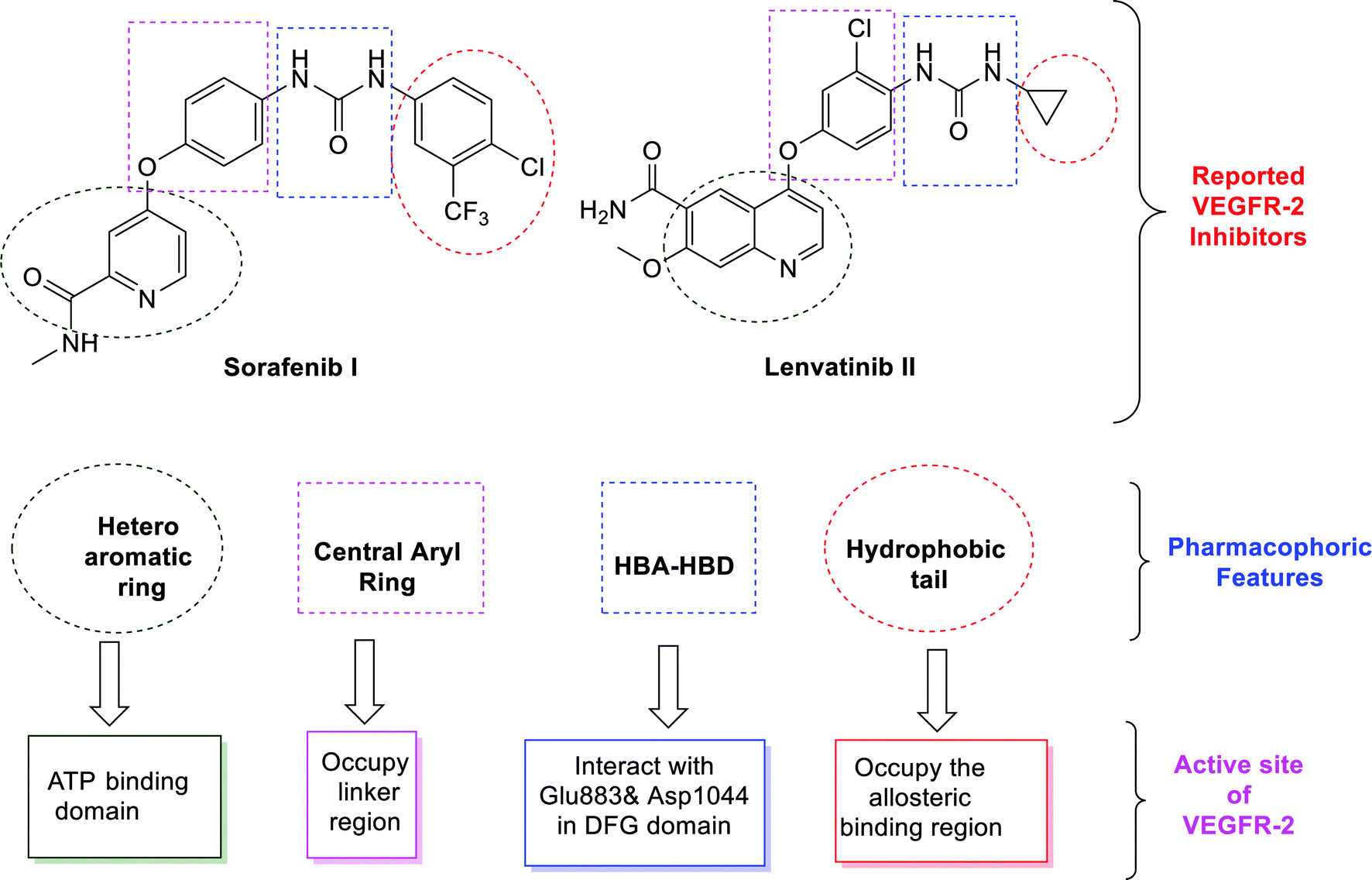
New quinoxaline-2(1 H )-ones as potential VEGFR-2 inhibitors: design, synthesis, molecular docking, ADMET profile and anti-proliferative evaluations - New Journal of Chemistry (RSC Publishing) DOI:10.1039/D1NJ02509K

Synthesis, antitumor and antimicrobial evaluation of novel 1,3,4-thiadiazole derivatives bearing disulfide bond | SpringerLink

Discovery of AXL Degraders with Improved Potencies in Triple-Negative Breast Cancer (TNBC) Cells | Journal of Medicinal Chemistry

Statistics of the Electrosensory Input in the Freely Swimming Weakly Electric Fish Apteronotus leptorhynchus | Journal of Neuroscience

Design, synthesis, and α‐glucosidase‐inhibitory activity of phenoxy‐biscoumarin–N‐phenylacetamide hybrids - Ansari - 2021 - Archiv der Pharmazie - Wiley Online Library

Synthesis of 2-(2-oxo-2H-chromen-3-yl)-3-phenylquinazolin-4(3H)-ones as potent antimicrobial and antitubercular agents - ScienceDirect

Biomolecules | Free Full-Text | Selenium-Substituted Monomethine Cyanine Dyes as Selective G-Quadruplex Spectroscopic Probes with Theranostic Potential

Standing Height Estimation from Sitting Height Measurements in Adolescents in the Central Region of Kosovo | Request PDF
![Efficient N‐Alkyl Enamination of 3‐(Thiobenzoyl)pyrrolo[2,3‐b]quinoxalin‐2‐one, E/Z Isomerisation of the Reaction Products and Potential Fluorescent Sensors for Zinc(II) Ion - Ostrowska - 2012 - European Journal of Efficient N‐Alkyl Enamination of 3‐(Thiobenzoyl)pyrrolo[2,3‐b]quinoxalin‐2‐one, E/Z Isomerisation of the Reaction Products and Potential Fluorescent Sensors for Zinc(II) Ion - Ostrowska - 2012 - European Journal of](https://chemistry-europe.onlinelibrary.wiley.com/cms/asset/88a420cd-ca7f-4f0e-80c0-698148c82968/mscheme1.jpg)
Efficient N‐Alkyl Enamination of 3‐(Thiobenzoyl)pyrrolo[2,3‐b]quinoxalin‐2‐one, E/Z Isomerisation of the Reaction Products and Potential Fluorescent Sensors for Zinc(II) Ion - Ostrowska - 2012 - European Journal of
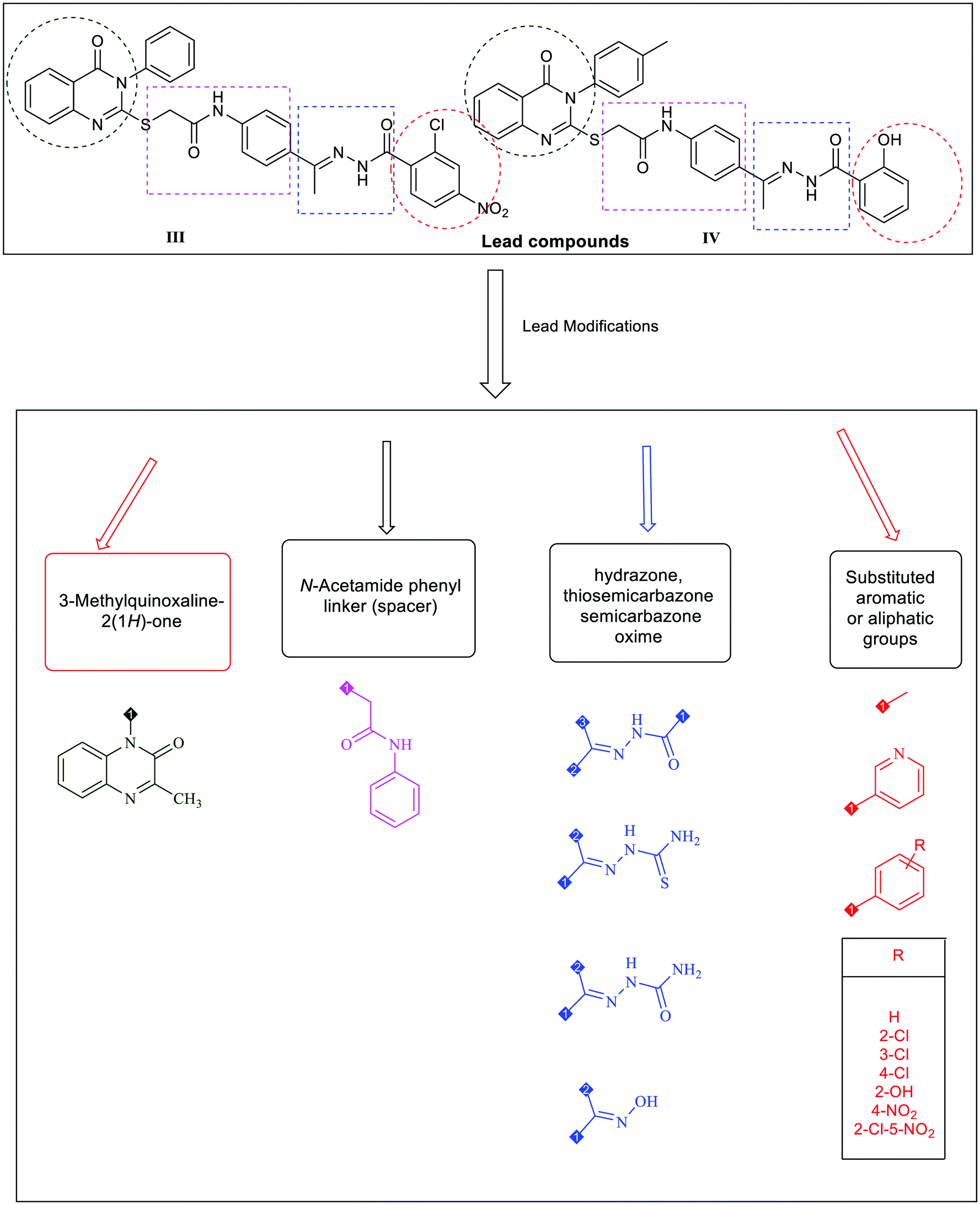
New quinoxaline-2(1 H )-ones as potential VEGFR-2 inhibitors: design, synthesis, molecular docking, ADMET profile and anti-proliferative evaluations - New Journal of Chemistry (RSC Publishing) DOI:10.1039/D1NJ02509K

Discovery of AXL Degraders with Improved Potencies in Triple-Negative Breast Cancer (TNBC) Cells | Journal of Medicinal Chemistry
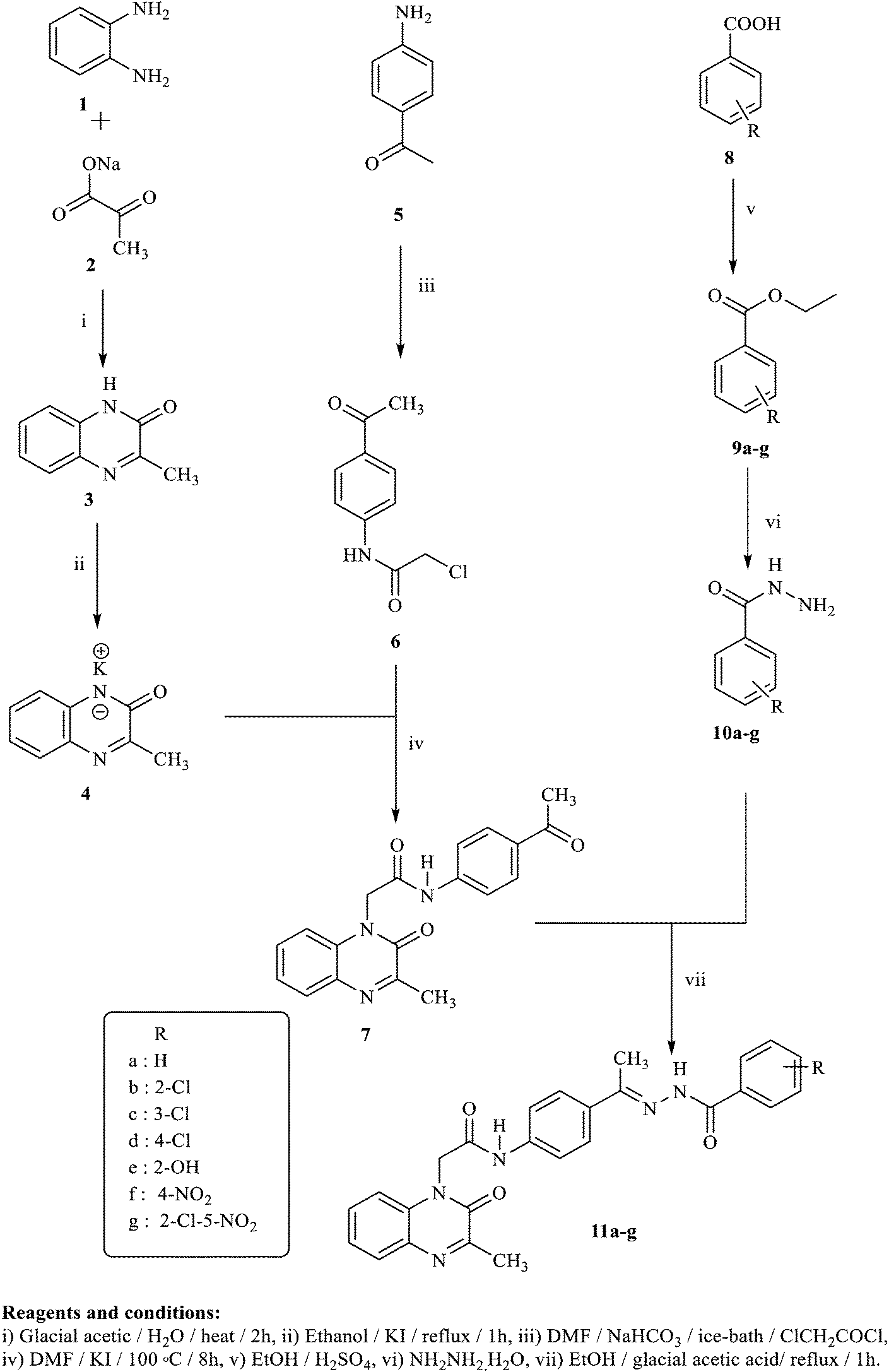
New quinoxaline-2(1 H )-ones as potential VEGFR-2 inhibitors: design, synthesis, molecular docking, ADMET profile and anti-proliferative evaluations - New Journal of Chemistry (RSC Publishing) DOI:10.1039/D1NJ02509K

Studies on the anticonvulsant activity of 4-alkyl-1,2,4-triazole-3-thiones and their effect on GABAergic system - ScienceDirect