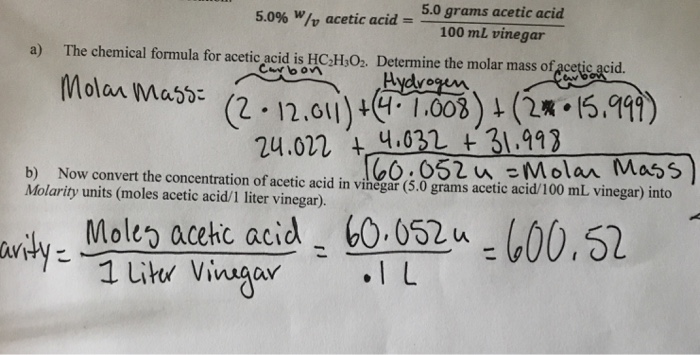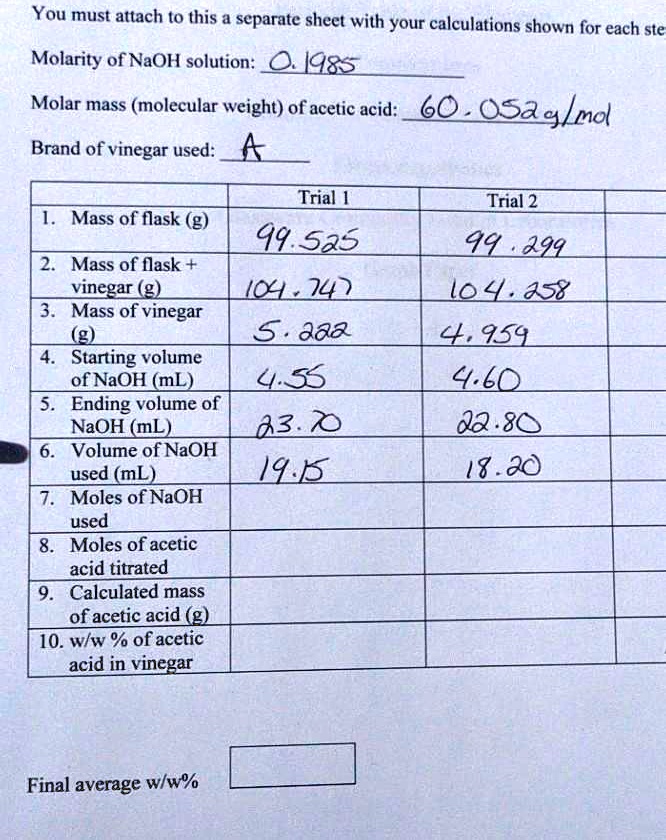
SOLVED: You must attach to this a separate shect with your calculations shown for each ste Molarity of NaOH solution: 14x5 Molar mass (molecular weight) of acetic acid: 60 OSag/md Brand of
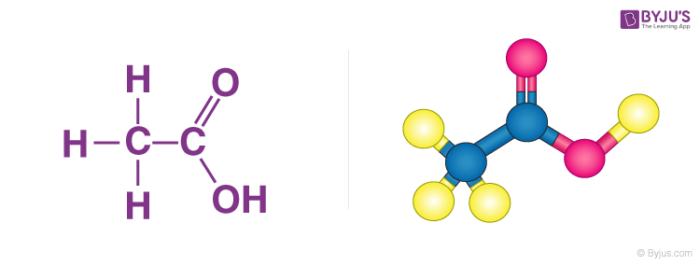
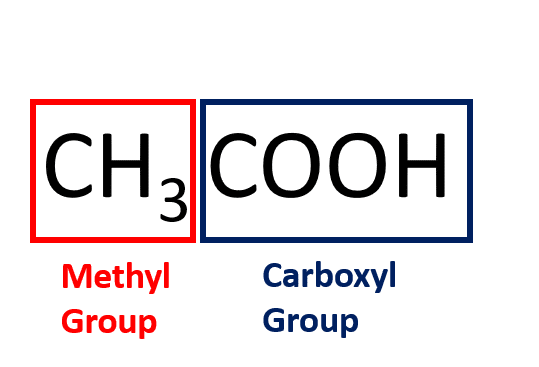
Acetic Acid (CH3COOH)- Structure, Properties, Preparation, Physical, Chemical properties, Uses and FAQs of Acetic acid

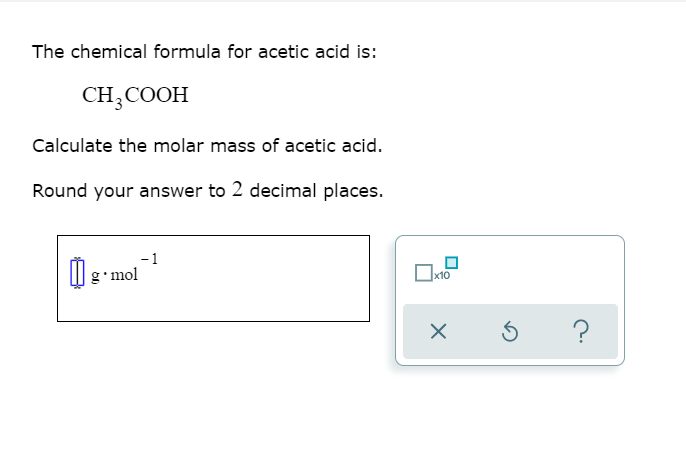
SOLVED: Question 3 1pts The molar mass of acetic acid, as determined with a mass spectrometer; is 60 g mol Given that the empirical (simplest) formula of acetic acid is CH2O,what is

Plan for Fri, 26 Sept 08 Diagnostic Quiz returned –Average = /- 18.1% Lecture –Naming acids and their anions (2.8) –Counting by weighing (3.1) –More. - ppt download

The experimental molecular weight of acetic acid is double the theoretical molecular weight of acetic acid. Why? - Quora