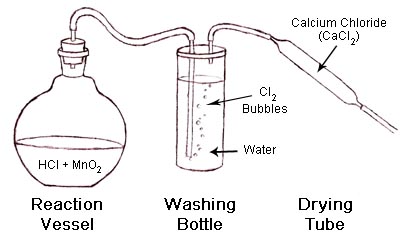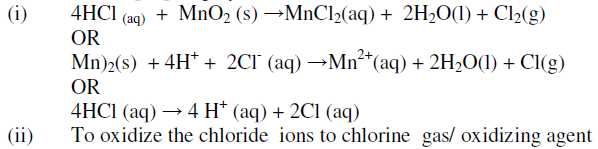
Chlorine gas is prepared by reacting concentrated hydrochloric acids with manganese (iv) oxide. i) Write the equation for reaction between concentrated hydrochloric acid and manganese (iv) oxide. ii)...

Chlorine is prepared in the laboratory by treating manganese dioxide(MnO2) with aqueous hydrochloric acid according to the reaction: 4HCl(aq) + MnO2(s)→ 2H2O(l) + MnCl2(aq) + Cl2(g) The amount of HCl that react
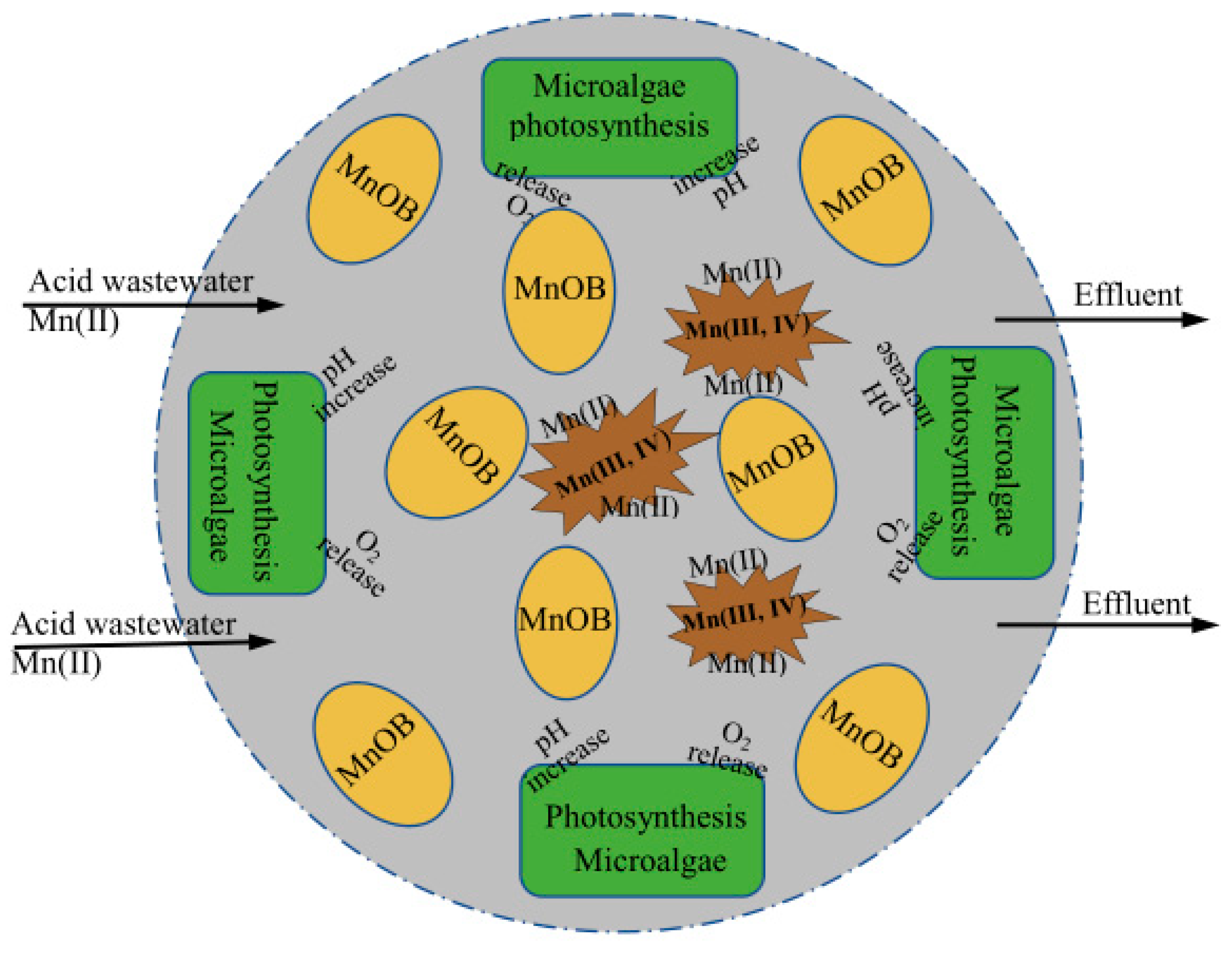
Water | Free Full-Text | Removal of Manganese(II) from Acid Mine Wastewater: A Review of the Challenges and Opportunities with Special Emphasis on Mn-Oxidizing Bacteria and Microalgae

SOLVED: The permanganate ion (Mn04'-) will react with chloride ions (Cl) to form manganese dioxide (MnOz) and chlorate ions (CIO3 in basic solution (40 pts) Balance the redox reaction in basic solution

Write the skeleton equations for the following chemical reactions and balance them by oxidation number method - CBSE Class 11 Chemistry - Learn CBSE Forum

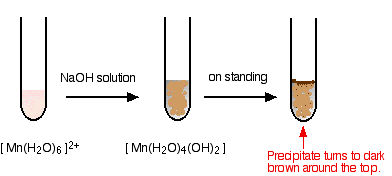
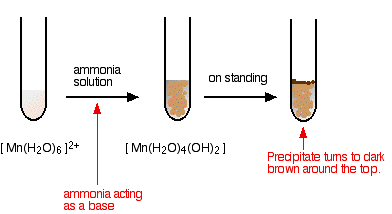
Manganese transition metal Chemistry manganese(II) Mn2+ complex ions MnO4- manganate(VII) 'permanganate' manganese(IV) oxide MnO2 redox chemical reactions principal oxidation states +2 +4 +6 +7 GCE AS A2 IB A level inorganic chemistry

Write the skeleton equations for the following chemical reactions and balance them by oxidation number method - CBSE Class 11 Chemistry - Learn CBSE Forum

Manganese transition metal Chemistry manganese(II) Mn2+ complex ions MnO4- manganate(VII) 'permanganate' manganese(IV) oxide MnO2 redox chemical reactions principal oxidation states +2 +4 +6 +7 GCE AS A2 IB A level inorganic chemistry

Write balanced chemical equation for the following reactions:a) Iodine ions can be oxidized by ozone in acidic medium.

Write the skeleton equation for the following chemical reaction and balance them by oxidation number method : (