
Pharmaceuticals | Free Full-Text | Identification of a Thyroid Hormone Derivative as a Pleiotropic Agent for the Treatment of Alzheimer’s Disease

PDF) A rationally designed bicyclic peptide remodels Aβ42 aggregation in vitro and reduces its toxicity in a worm model of Alzheimer's disease

Selective targeting of primary and secondary nucleation pathways in Aβ42 aggregation using a rational antibody scanning method | Science Advances

PDF) Delivery of Native Proteins into C. Elegans Using a Transduction Protocol Based on Lipid Vesicles
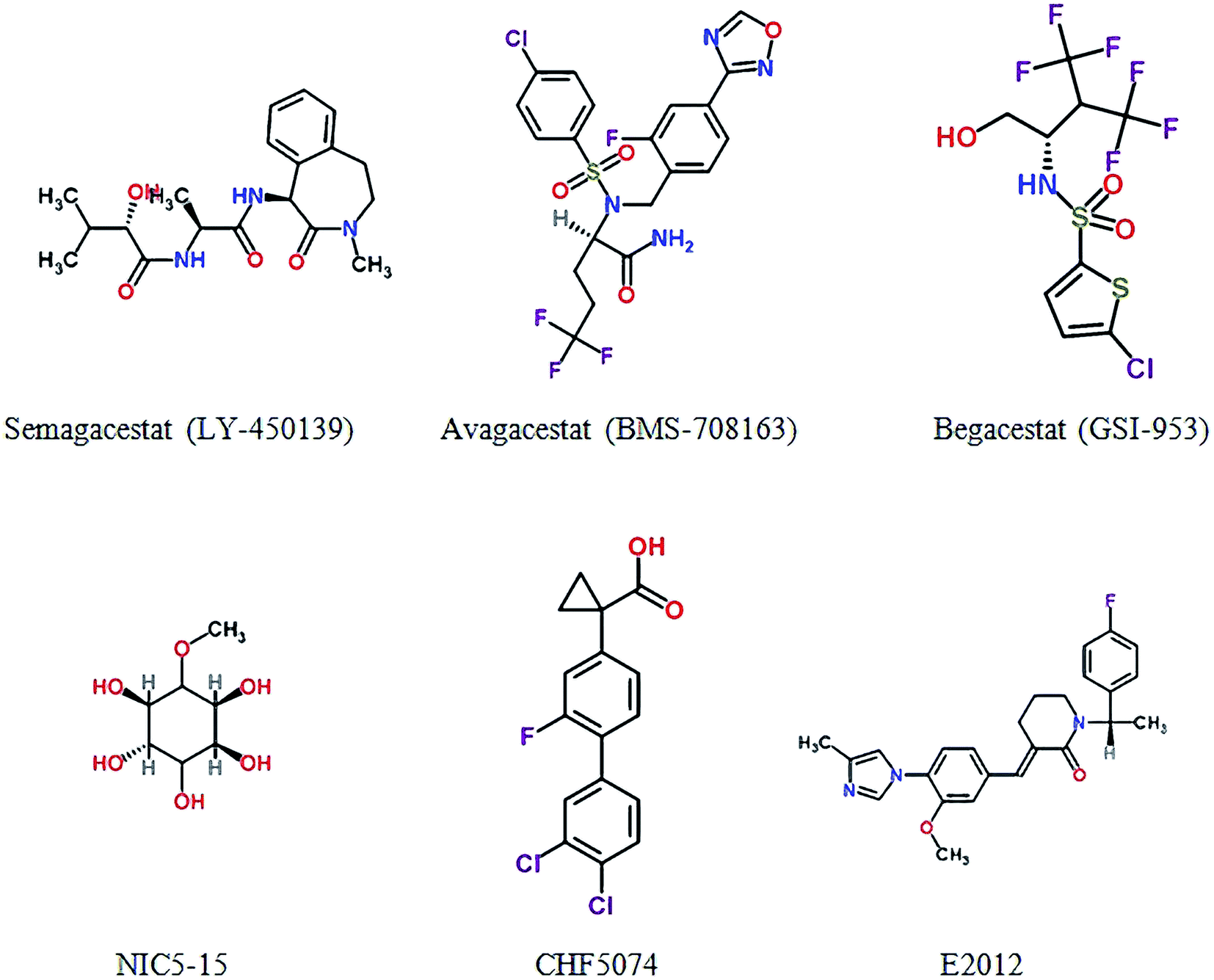
Two decades of new drug discovery and development for Alzheimer's disease - RSC Advances (RSC Publishing) DOI:10.1039/C6RA26737H

Drug discovery: Insights from the invertebrate Caenorhabditis elegans - Giunti - 2021 - Pharmacology Research & Perspectives - Wiley Online Library

An anticancer drug suppresses the primary nucleation reaction that initiates the production of the toxic Aβ42 aggregates linked with Alzheimer's disease | Science Advances

PDF) Systematic development of small molecules to inhibit specific microscopic steps of Aβ42 aggregation in Alzheimer's disease

An anticancer drug suppresses the primary nucleation reaction that initiates the production of the toxic Aβ42 aggregates linked with Alzheimer's disease | Science Advances

Perphenazine–Macrocycle Conjugates Rapidly Sequester the Aβ42 Monomer and Prevent Formation of Toxic Oligomers and Amyloid | ACS Chemical Neuroscience

Rational design of a conformation-specific antibody for the quantification of Aβ oligomers. - Abstract - Europe PMC

An anticancer drug suppresses the primary nucleation reaction that initiates the production of the toxic Aβ42 aggregates linked with Alzheimer's disease | Science Advances

PDF) Selective targeting of primary and secondary nucleation pathways in Aβ42 aggregation using a rational antibody scanning method
![Hexahydropyrrolo[2,3-b]indole Compounds as Potential Therapeutics for Alzheimer's Disease | ACS Chemical Neuroscience Hexahydropyrrolo[2,3-b]indole Compounds as Potential Therapeutics for Alzheimer's Disease | ACS Chemical Neuroscience](https://pubs.acs.org/cms/10.1021/acschemneuro.9b00297/asset/images/medium/cn9b00297_0007.gif)
Hexahydropyrrolo[2,3-b]indole Compounds as Potential Therapeutics for Alzheimer's Disease | ACS Chemical Neuroscience

Rational design of a conformation-specific antibody for the quantification of Aβ oligomers. - Abstract - Europe PMC

PDF) Exogenous misfolded protein oligomers can cross the intestinal barrier and cause a disease phenotype in C. elegans
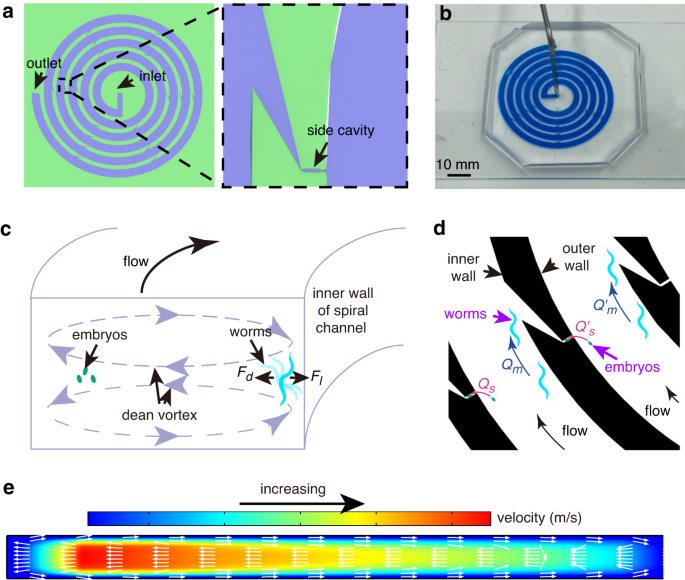
A spiral microfluidic device for rapid sorting, trapping, and long-term live imaging of Caenorhabditis elegans embryos | Microsystems & Nanoengineering

C-Terminal Fragment, Aβ39–42-Based Tetrapeptides Mitigates Amyloid-β Aggregation-Induced Toxicity | ACS Omega

An anticancer drug suppresses the primary nucleation reaction that initiates the production of the toxic Aβ42 aggregates linked with Alzheimer's disease | Science Advances

Genetic and Pharmacological Discovery for Alzheimer's Disease Using Caenorhabditis elegans | ACS Chemical Neuroscience
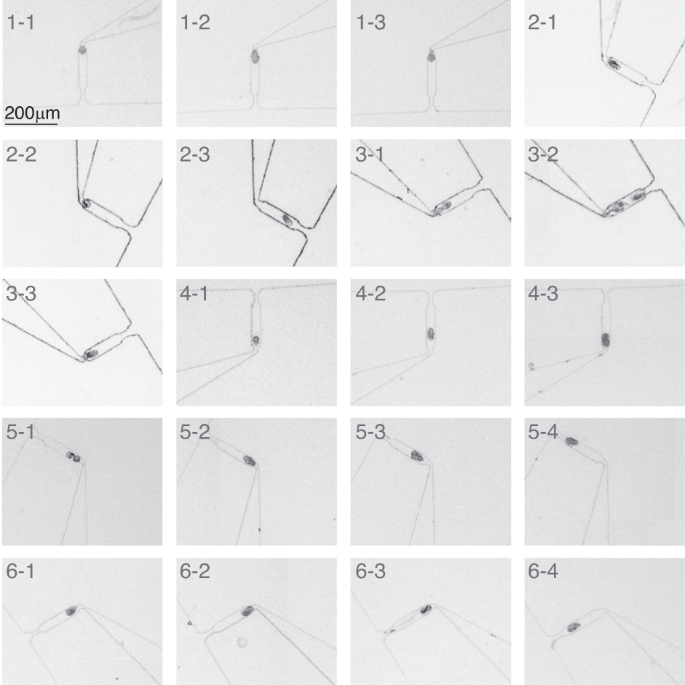
A spiral microfluidic device for rapid sorting, trapping, and long-term live imaging of Caenorhabditis elegans embryos | Microsystems & Nanoengineering



