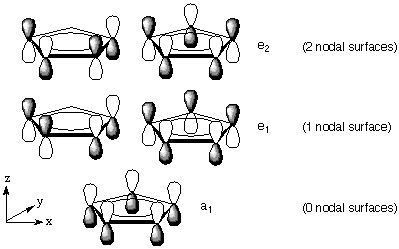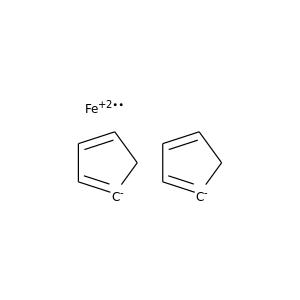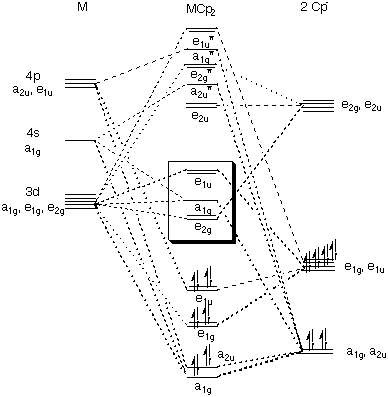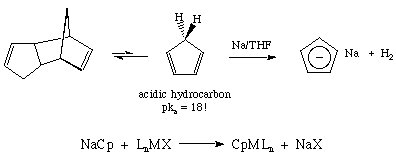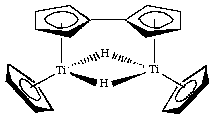
Cross-coupling of alkyl halides with aryl or alkyl Grignards catalyzed by dinuclear Ni(ii) complexes containing functionalized tripodal amine-pyrazolyl ligands - Dalton Transactions (RSC Publishing)

Dimerization of Terminal Aryl Alkynes Catalyzed by Iron(II) Amine-Pyrazolyl Tripodal Complexes with <i>E</i>/<i>Z</i> Selectivity Controlled by <i>tert</i>-Butoxide. - Abstract - Europe PMC

Fe Organoiron Compounds: Mononuclear Compounds 11 (Gmelin Handbook of Inorganic and Organometallic Chemistry - 8th edition, F-e / A-C / B / 11): Krüerke, Ulrich, Drechsler, Alfred, Rudolph, Edgar: 9783662069172: Amazon.com: Books

PDF) Iron Catalysis in Organic Synthesis: A Critical Assessment of What It Takes To Make This Base Metal a Multitasking Champion

Dimerization of Terminal Aryl Alkynes Catalyzed by Iron(II) Amine-Pyrazolyl Tripodal Complexes with <i>E</i>/<i>Z</i> Selectivity Controlled by <i>tert</i>-Butoxide. - Abstract - Europe PMC

Recent advance of chemoenzymatic catalysis for the synthesis of chemicals: Scope and challenge - ScienceDirect

Organic Chemistry of Transition Metals. I. New Reactions of Bis arene Iron II Salts. II. Synthesis and Structure of Pi-Cyclopentadienyl-Pi-Tetra-Phenylcyclobutadienerhodium i: Amazon.de: Cash, Gordon Graham: Fremdsprachige Bücher
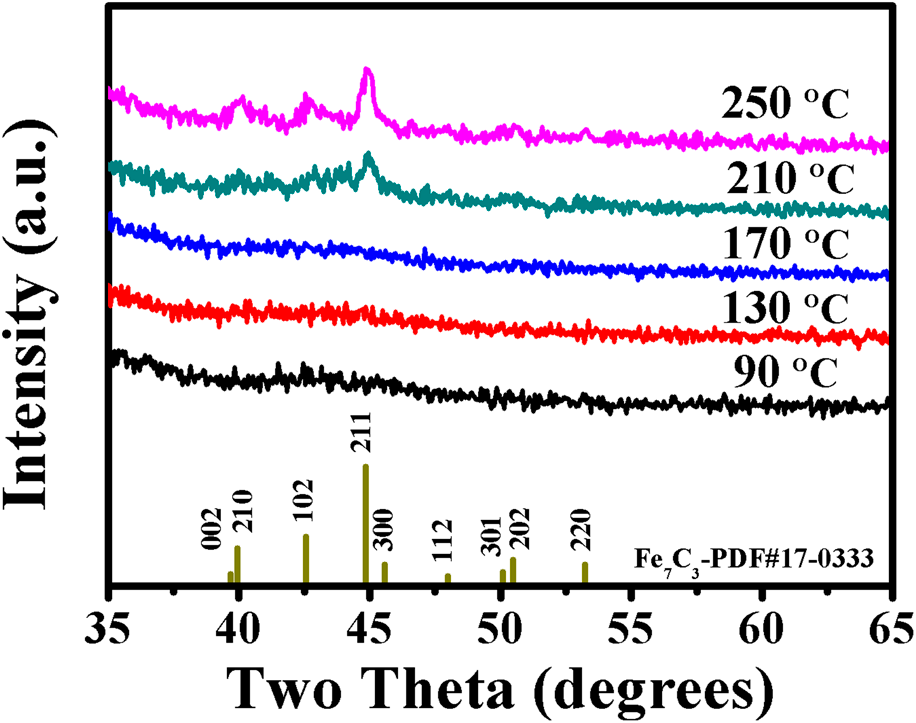
Fabrication of iron carbide by plasma-enhanced atomic layer deposition | Journal of Materials Research | Cambridge Core

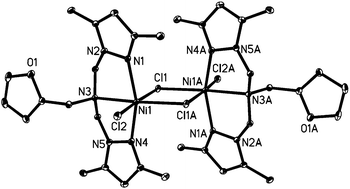
PDF superior Cadmium and mercury complexes containing trinuclear titanium imido nitrido metalloligands - 1Library.Co

Fabrication of iron carbide by plasma-enhanced atomic layer deposition | Journal of Materials Research | Cambridge Core
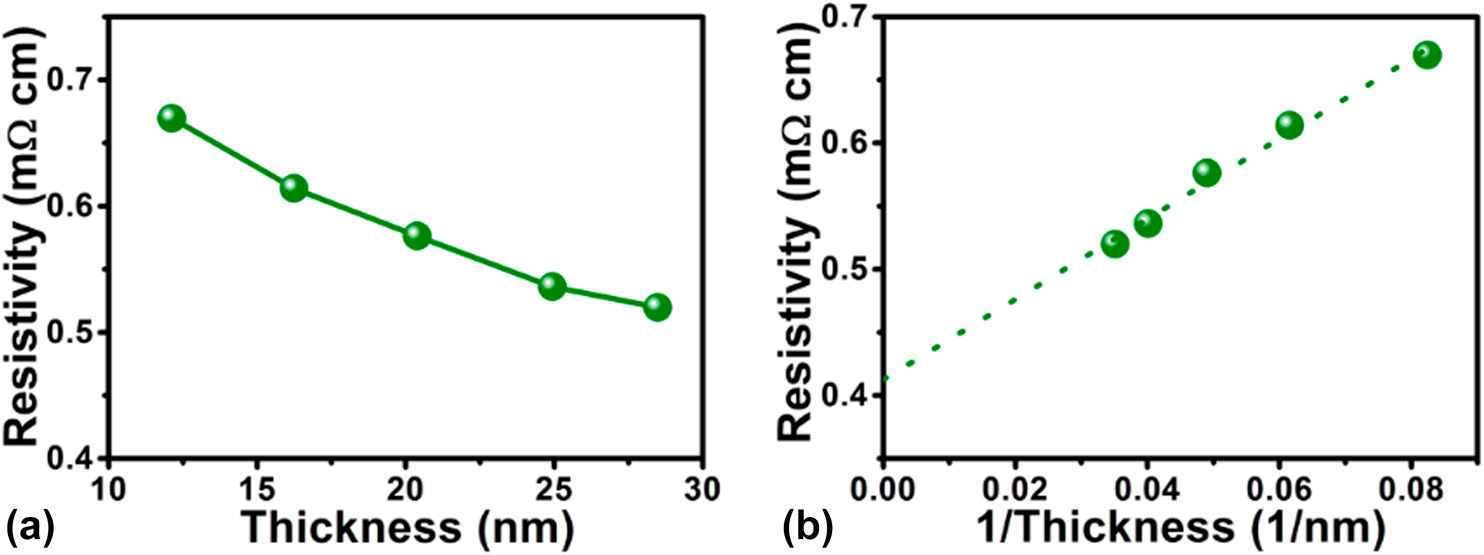
Fabrication of iron carbide by plasma-enhanced atomic layer deposition | Journal of Materials Research | Cambridge Core

A triplet biradical organocalcium species derived from partial oxygen quenching of a substituted cyclopentadienyl calcium compound | Gardiner, Michael G.; Hanson, Graeme R.; Junk, Peter C.; Raston, Colin L.; Skelton, Brian W.;