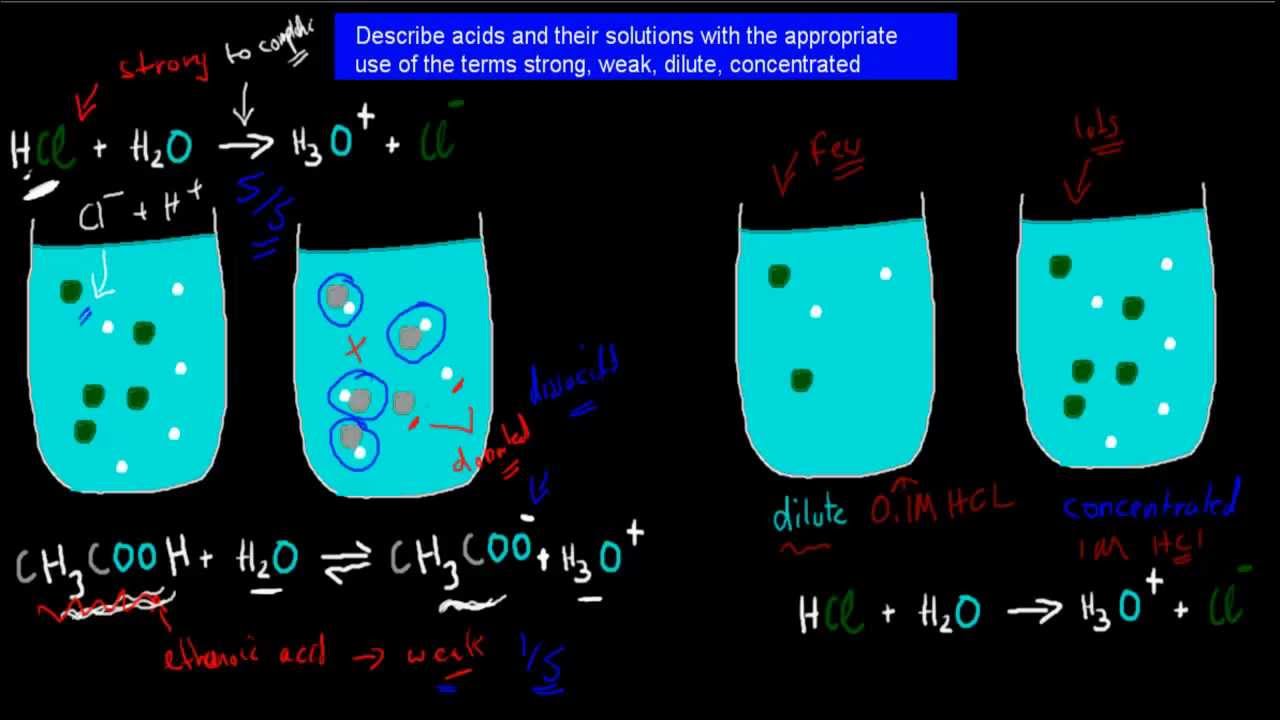
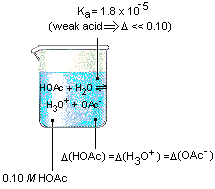Dan Beech 在Twitter: "Modelling strong & weak acids using lego as suggested by @RSC_EiC @BCAScience #dilute #concentrated #strong #weak https://t.co/xexYRuvZJD" / Twitter

SOLVED: Assertion-On dilution of a strong acid or a weak acid the number of H+ ions per unit volume in the solution decreases. Reason-dissociation of strong acid does not increase with dilution
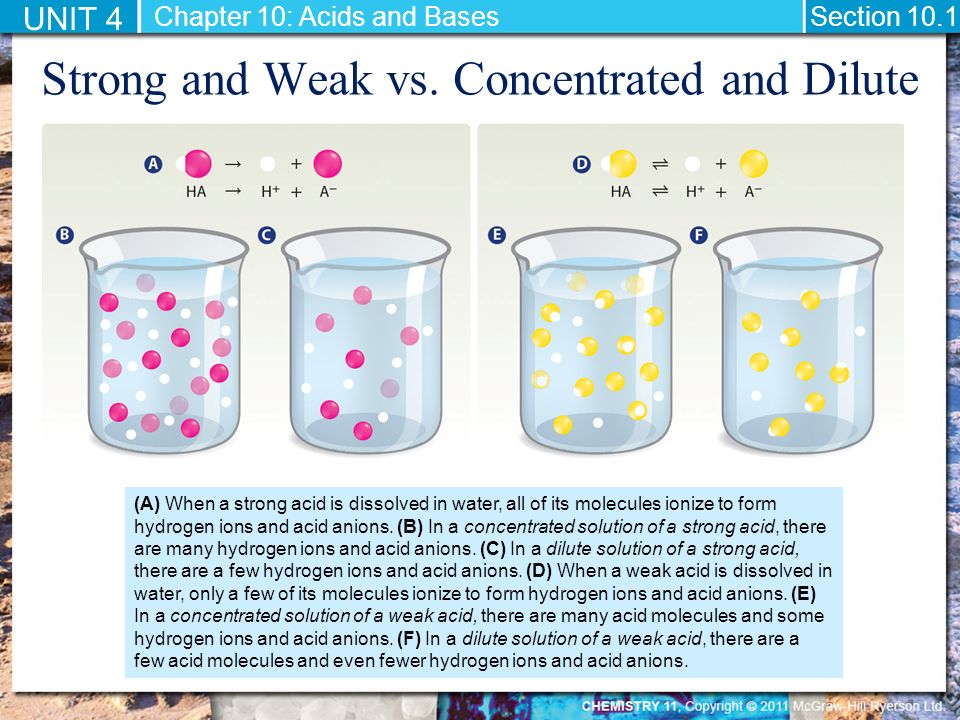
UNIT 4: Solutions: Dilutions & Titrations. Strong Acids An acid that ionizes completely in water is called a strong acid. Hydrochloric acid, HCl(aq), - ppt download

0.04 N` solution of a weak acid has specific conductance `4.23 xx 10^(-4) "mho cm"^(-1)`. If the - YouTube

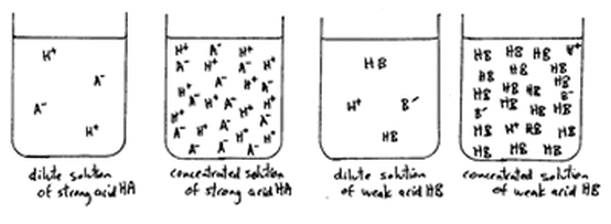
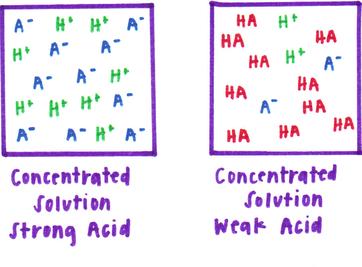
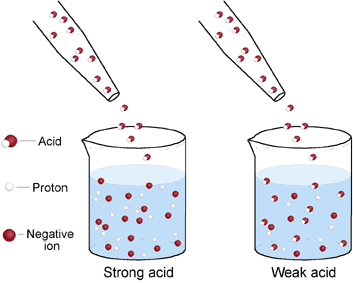
HSC Chemistry : Models of Strong, Weak, Concentrated and Dilute Acid and Bases - Art Of Smart Education