Halide Anion Activated Reactions of Michael Acceptors with Tropylium Ion | Organic Chemistry | ChemRxiv | Cambridge Open Engage

Impact of Chemically Specific Interactions between Anions and Weak Polyacids on Chain Ionization, Conformations, and Solution Energetics | Macromolecules

Anion Specific Effects at Negatively Charged Interfaces: Influence of Cl–, Br–, I–, and SCN– on the Interactions of Na+ with the Carboxylic Acid Moiety | The Journal of Physical Chemistry B
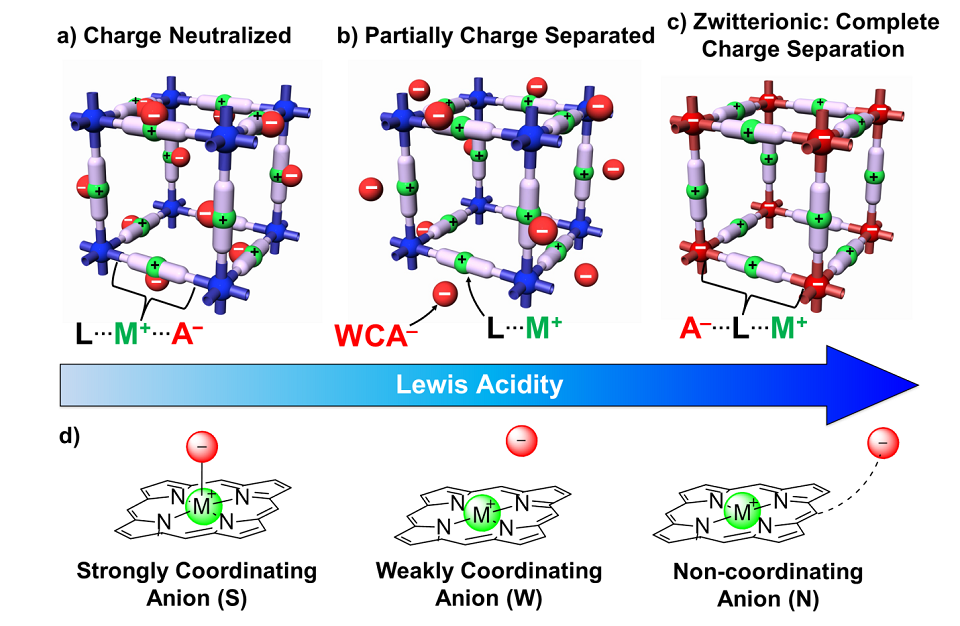
A New Approach to Non-Coordinating Anions for Electrocyclization Reactions | Department of Chemistry

SOLVED: For the following three anions: a) Give the hybridization of the atomic orbital containing the non-bonding electrons and draw a picture of its wavefunction on top of each anion. b) Rank










![PDF] Treatment of acute non-anion gap metabolic acidosis | Semantic Scholar PDF] Treatment of acute non-anion gap metabolic acidosis | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/3e74f8c1cc31130408db1e788c3b32fb25586e8e/5-Figure1-1.png)