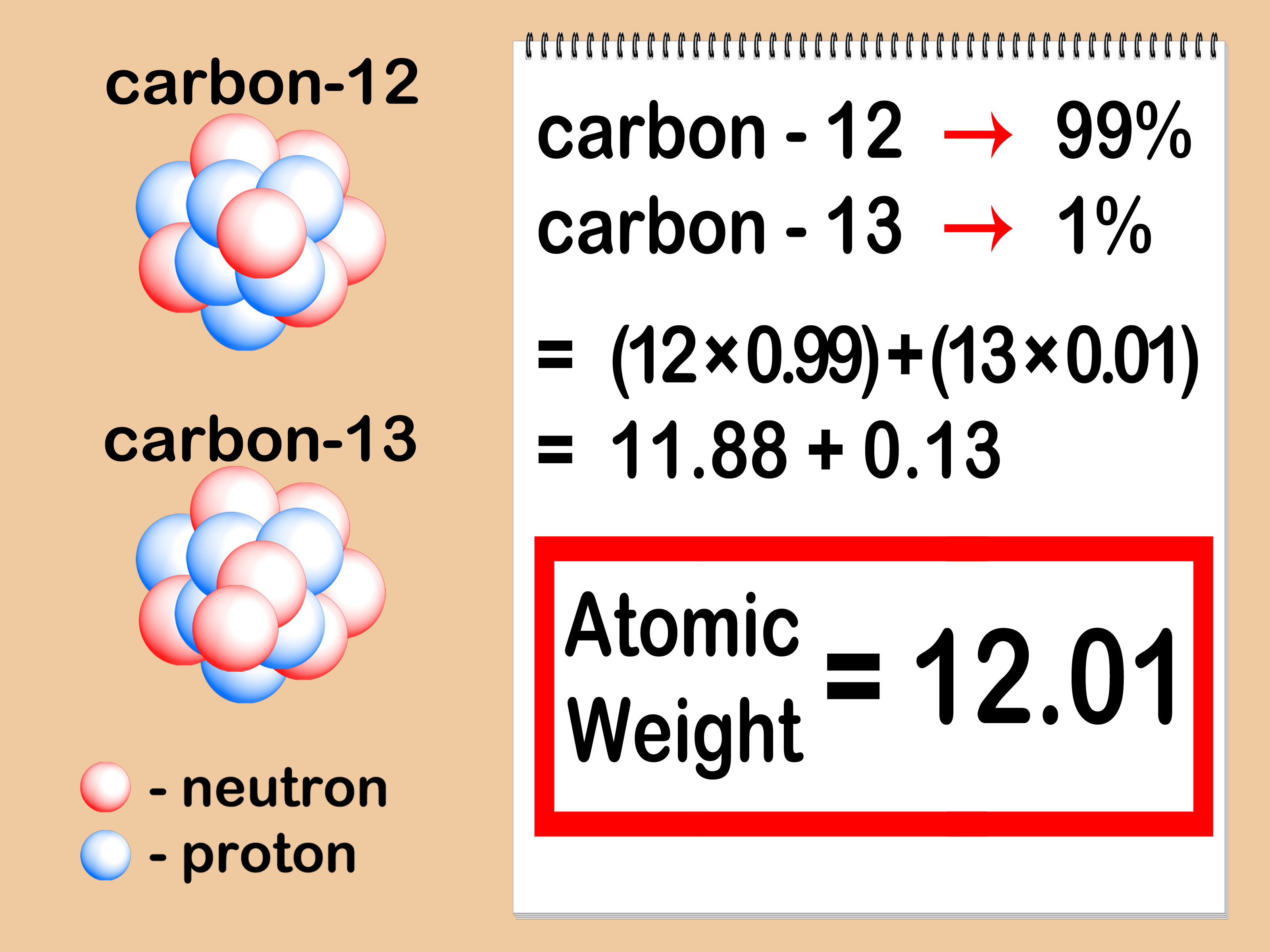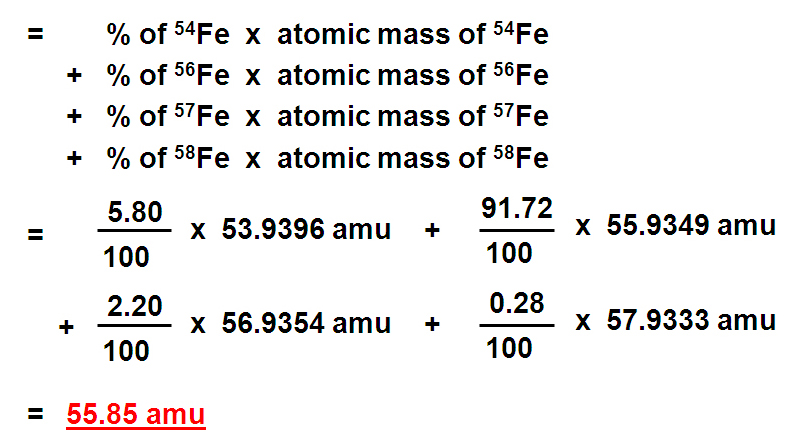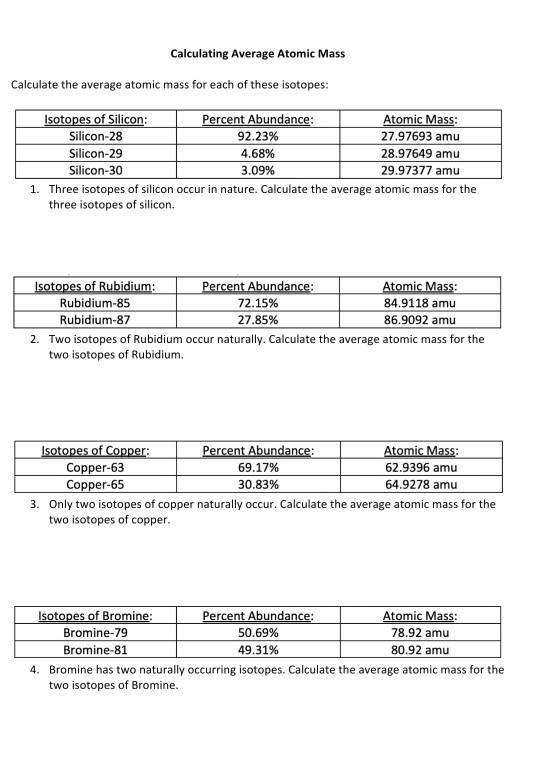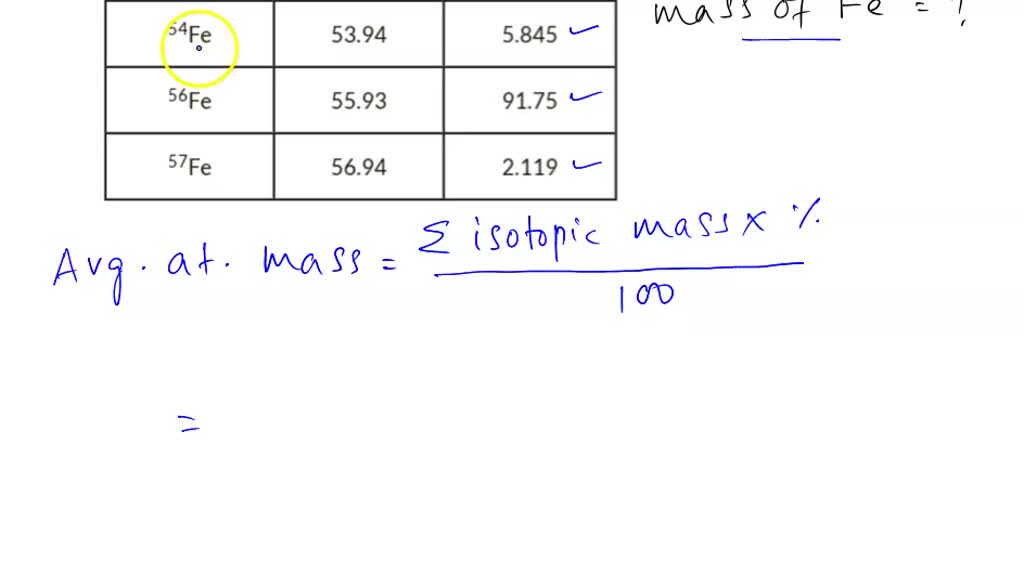
SOLVED: Calculate the average atomic mass of iron using the isotopes in Table 2 below: Table 2. Stable Isotopes of Iron Isotope Mass (amu) % Abundance 54Fe 53.94 5.845 56Fe 55.93 91.75 57Fe 56.94 2.119

An element has average atomic mass 20.426u it has 2 isotopes with masses 22.1824 and 19.9574 . Calculate the abundance of its isotopes X - 22 and X - 20 .