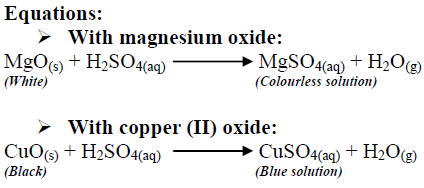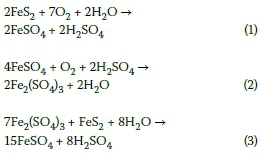THE RICO-ARGENTINE CO. SULPHURIC ACID MILL. ALTHOUGH THE MILL HAS BEEN CLOSED FOR SIX YEARS, WATER SEEPING FROM OLD MINE SHAFTS POLLUTES NEARBY STREAMS WITH IRON OXIDE Stock Photo - Alamy

Fe2O3+H2SO4=Fe2(SO4)3+H2O Balanced Equation|| Balanced equation for Iron iii oxide and Sulfuric acid - YouTube

WATER SEEPING FROM CLOSED MINE CARRIES IRON OXIDE AND OTHER MINERALS INTO NEARBY STREAMS. THE MINE BELONGS TO THE RICO-ARGENTINE MILL, WHICH PRODUCED SULPHURIC ACID. THE PLANT WAS SHUT DOWN SIX YEARS

Calculate the equivalent mass of sulphuric acid (ii) The reaction between aluminium and ferric oxide can generate temperatures up to 3273 K and is used in welding metals. (Atomic mass of AI=

Sulfuric Acid LO: Outline uses and reactions involving Sulfuric Acid Starter: What is an acid? - ppt download

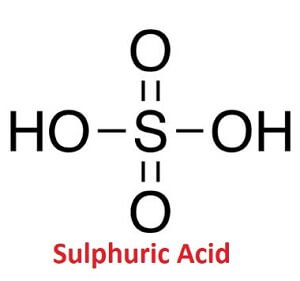
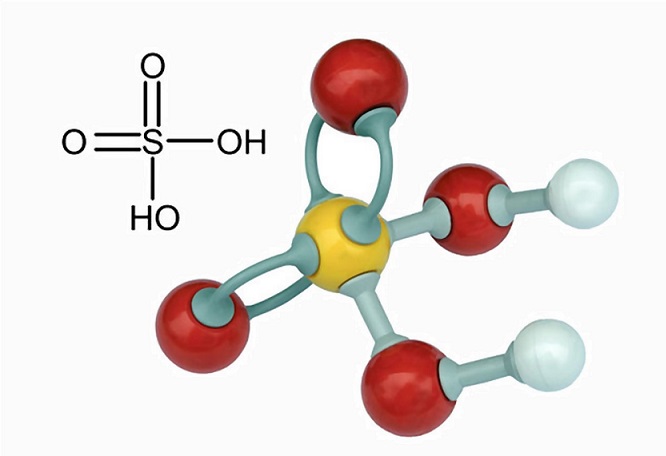
Write word equations and then balanced equations for the reaction taking place when:(a) Dilute sulphuric acid reacts with zinc granules.(b) Dilute hydrochloric acid reacts with magnesium ribbon.(c) Dilute sulphuric acid reacts with

The Oxidation of Fe(II) in Acidic Sulfate Solutions with Air at Elevated Pressures. Part 1. Kinetics above 1 M H2SO4 | Industrial & Engineering Chemistry Research

Calculate the equivalent mass of sulphuric acid (ii) The reaction between aluminium and ferric oxide can generate temperatures up to 3273 K and is used in welding metals. (Atomic mass of AI=

Explain with the help of chemical equations what happens when: - (a) Copper is heated strongly in air.(b) Iron is reached with steam.(c) Sodium is reacted with dilute sulphuric acid.(d) Zinc is

A Facile One‐Pot Transformation of Aromatic Aldehydes/Ketones to Amides: Fe2O3@SiO2 as an Environmentally Benign Core‐Shell Catalyst - Jain - 2018 - ChemistrySelect - Wiley Online Library

SOLVED: 'please answer these questions please 9,10 and 11 0 Explain why sulphuric acid forms two types of salts. 10. Give balanced equations for the preparation of the following salts: Calcium oxide

The process of pyrite weathering in a deep metal mine. Four general... | Download Scientific Diagram

7.2 Making Salts What is made when acids and metals react? What is made when alkalis and metals react? 27 August 2015 Bonneville salt flats in America. - ppt download

balance the equation :- ferric oxide reacts with sulphuric acid and give ferric sulphate and water. - Brainly.in