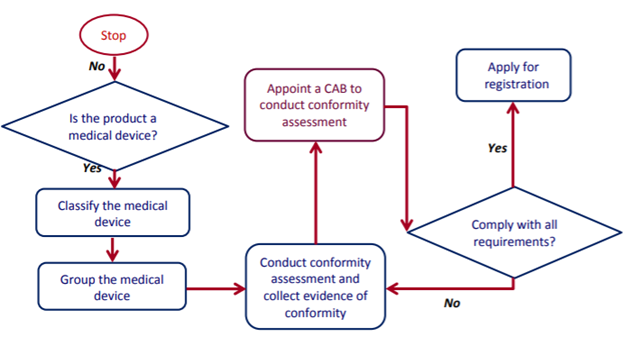
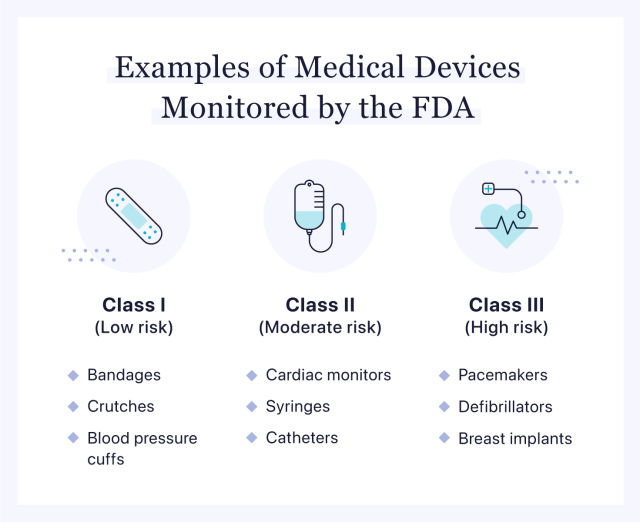
Medical Device Registration: A Guide For Importers - Life Sciences, Biotechnology & Nanotechnology - India

Ministry of Food and Drug Safety>Information>News&Notice>Card News | Ministry of Food and Drug Safety

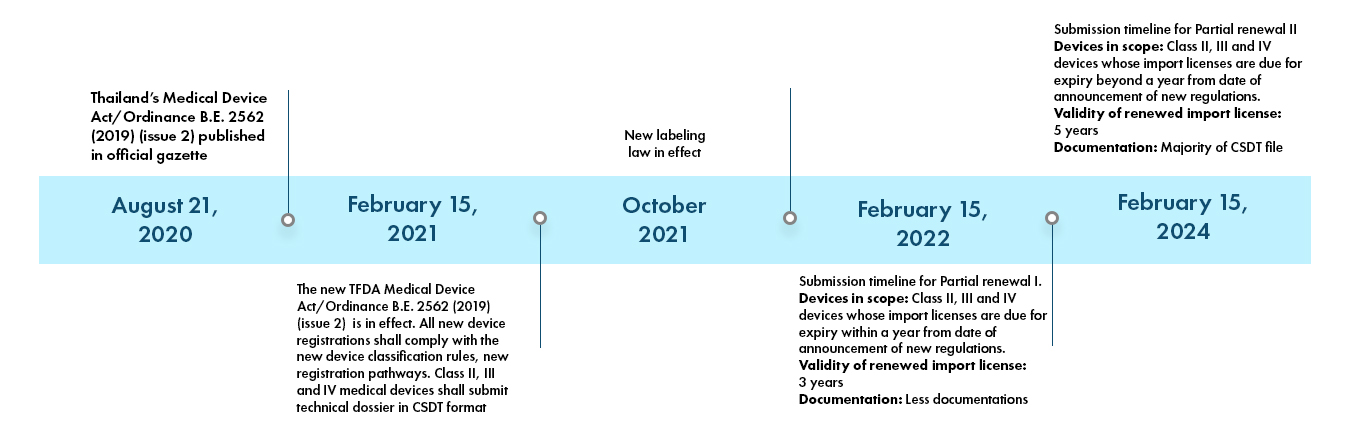
Adapting to Taiwan's New Medical Device Act: Navigating Changes, Execution Dates, and Regulatory Challenges for Industry Players
Unofficial Translation MEDICAL DEVICES ACT, B.E. 2551 (2008) ______ BHUMIBOL ADULYADEJ, REX. Given on the 26 Day of Febru