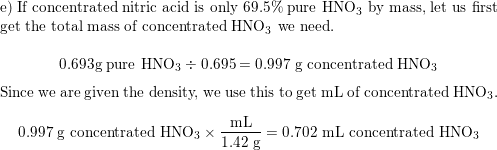The pH titration curves of nitric acid (C 0 a = 0.0002 mol l −1 ) being... | Download Scientific Diagram
![PDF] USE OF NITRIC ACID IN CONTROL OF pH AND NITRATE LEVELS IN NUTRIENT SOLUTION. | Semantic Scholar PDF] USE OF NITRIC ACID IN CONTROL OF pH AND NITRATE LEVELS IN NUTRIENT SOLUTION. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/60b90fc2a9b563e870ebeafd75799efd7933897d/1-TableI-1.png)
PDF] USE OF NITRIC ACID IN CONTROL OF pH AND NITRATE LEVELS IN NUTRIENT SOLUTION. | Semantic Scholar
![SOLVED:A nitric acid solution is found to have a pH of 2.70. Determine each of the following: a. [H3 O^+] b. [OH^-] c. the number of moles of HNO 3 required to SOLVED:A nitric acid solution is found to have a pH of 2.70. Determine each of the following: a. [H3 O^+] b. [OH^-] c. the number of moles of HNO 3 required to](https://cdn.numerade.com/previews/7385785b-c06a-42ad-bf7a-33b53fdc7fee_large.jpg)
SOLVED:A nitric acid solution is found to have a pH of 2.70. Determine each of the following: a. [H3 O^+] b. [OH^-] c. the number of moles of HNO 3 required to

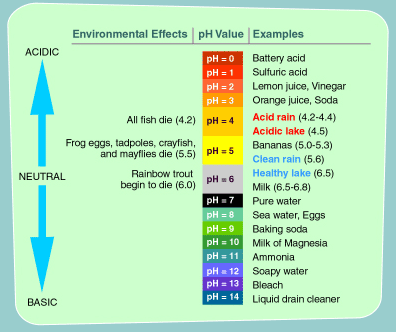
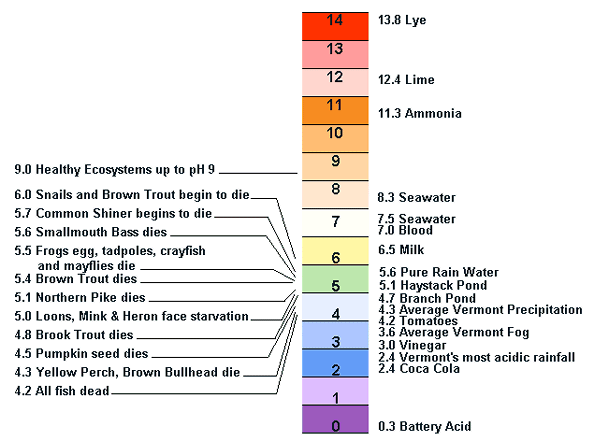
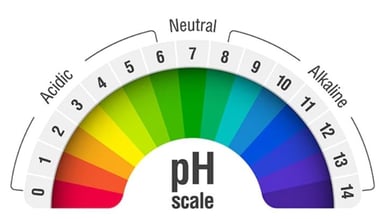
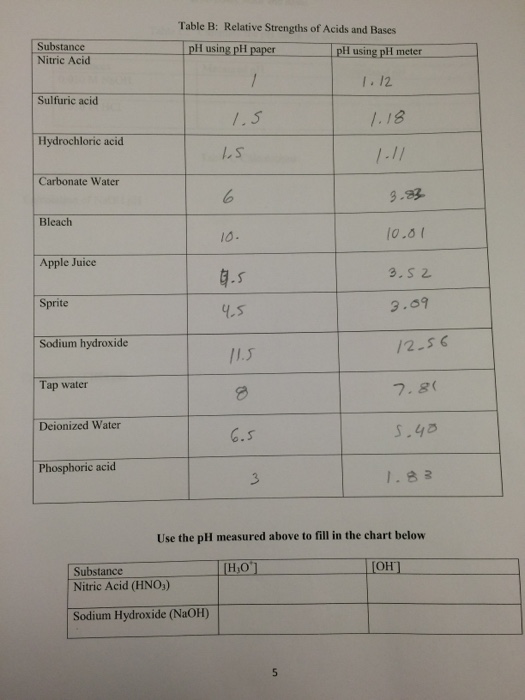
What is pH? | College of Agriculture, Forestry and Life Sciences | Clemson University, South Carolina

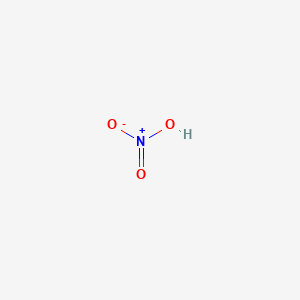
A solution was prepared by dissolving 0.63g of nitric acid in 10 L of the solution. Calculate its pH.