science chemistry titration potassium permanganate sodium dichromate | Fundamental Photographs - The Art of Science
Which is the type of reaction of potassium permanganate and oxalic acid: endothermic or exothermic? - Quora

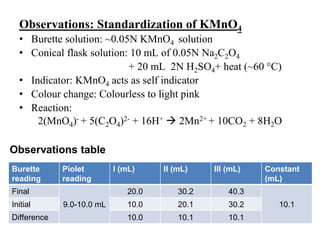
Determination of concentration of KMnO₄ solution (Theory) : Class 12 : Chemistry : Online Labs for schools - Developed by Amrita Vishwa Vidyapeetham and CDAC Online Lab

what is n factor of KmO4 when it react with oxalic acid - Chemistry - Some Basic Concepts of Chemistry - 13663621 | Meritnation.com
Prepare N/20 solution of oxalic acid. Using this solution, find out strength and normality of the given potassium permanganate solution

Standard oxalic acid concentration vs. burette readings i.e. ml of KMnO... | Download Scientific Diagram

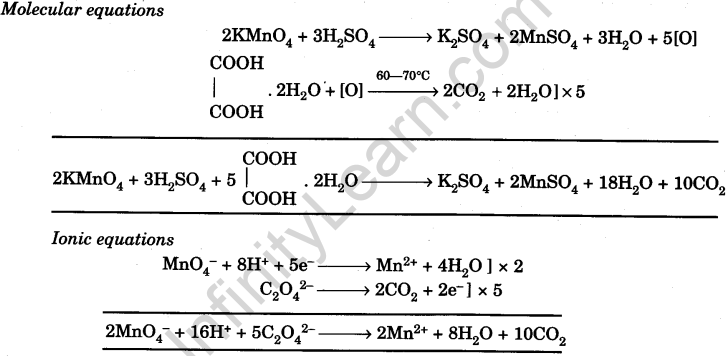
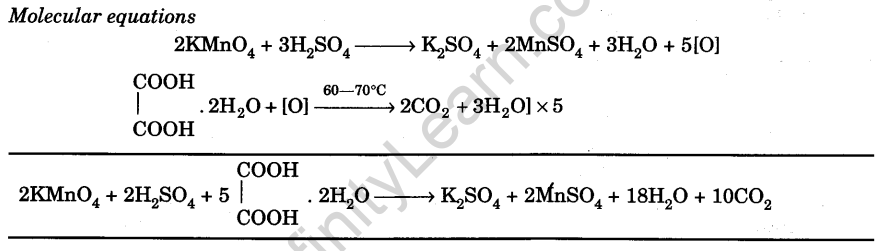
KMnO4 reacts with oxalic acid according to the equation, 2MnO ^ - 4 + 5C2O^2 - 4 + 16H^ + → 2Mn^2 + + 10CO2 + 8H2O , here 20 ml of 0.1 M KMnO4 is equivalent to:

Explain why does color of KMnO4 disappear when oxalic acid is added to its solution in acidic medium.

OneClass: Potassium permanganate, KMnO4, reacts with oxalic acid, H2C2O4, in aqueous sulfuric acid ac...
KMnO4 reacts with oxalic acid according to the equation - Sarthaks eConnect | Largest Online Education Community

KMnO4 reacts with oxalic acid according to the equation: 2MnO4^- + 5C2O4^2 - + 16H^+→ 2Mn^2 + + 10CO2 + 8H2O Here, 20 mL of 0.1 M KMnO4 is equivalent to:
Prepare M/50 Solution of Oxalic Acid. With its help, Determine 50 the Molarity and Strength of the Given Solution of Potassium Permanganate - Infinity Learn