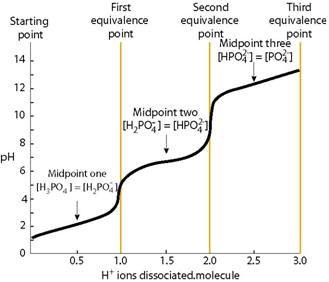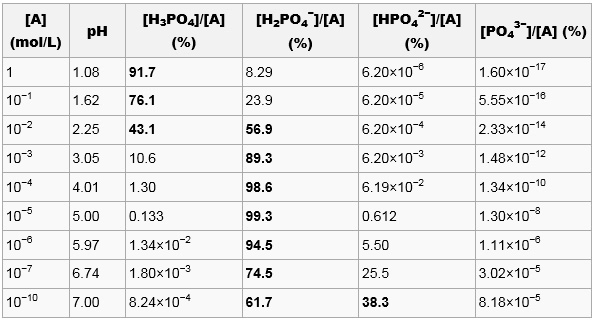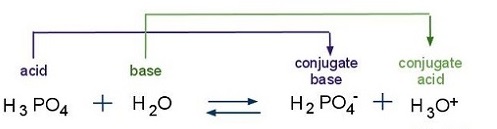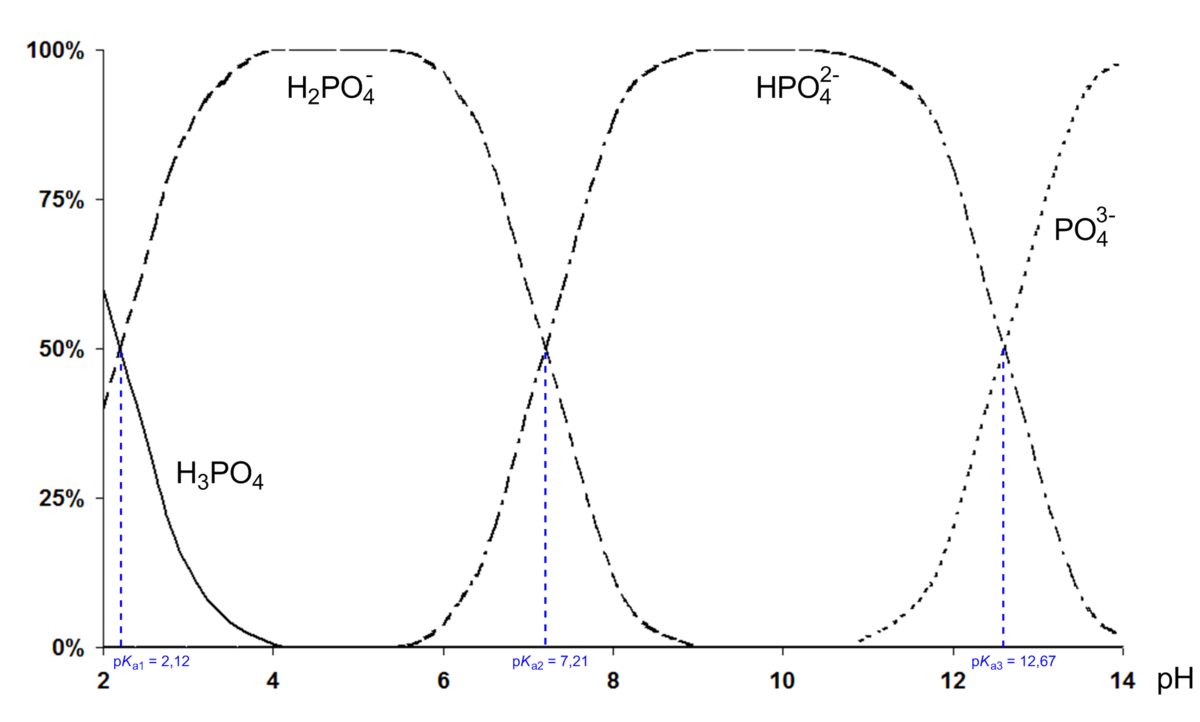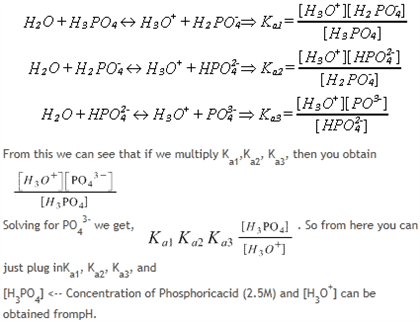
OneClass: What is the concentration of phosphate ions in a 2.5 M solution ofphosphoric acid?(The pH o...

Determination of the Apparent Dissociation Constants of Phosphoric Acid in SEAWATER1 | Semantic Scholar

SOLVED: Phosphoric acid, H3PO4 is a triprotic acid. pKa1= 2.16 pKa2=7.21 pKa3=12.32 Write three separate acid dissociation reactions for the three acidic protons. Make sure to indicate which Ka and Kb value

SOLVED: Acid dissociation constants for phosphoric acid are given below. H3PO4 Ka1=7.5*10^-3 Ka2=6.2*10^-8 Ka3=4.8*10^-13 A buffer with a pH = 7.4 can be best be made by using? Fill in the answer below

The values for pkas are given, can someone tell me which one corresponds to which form of the phosphoric acid? Thanks! : r/Mcat