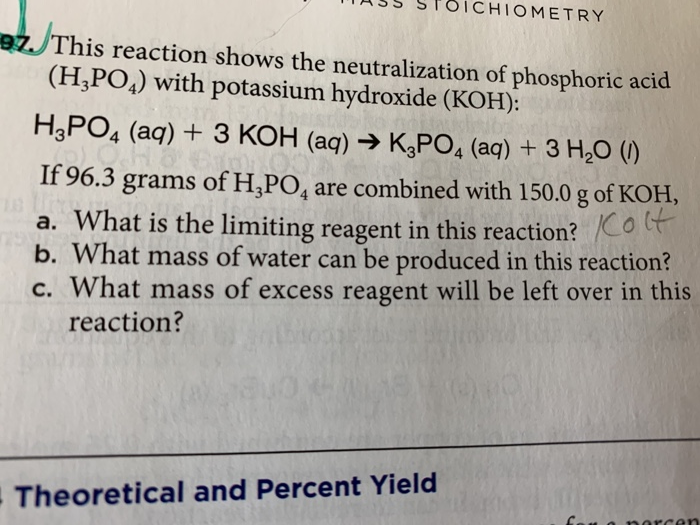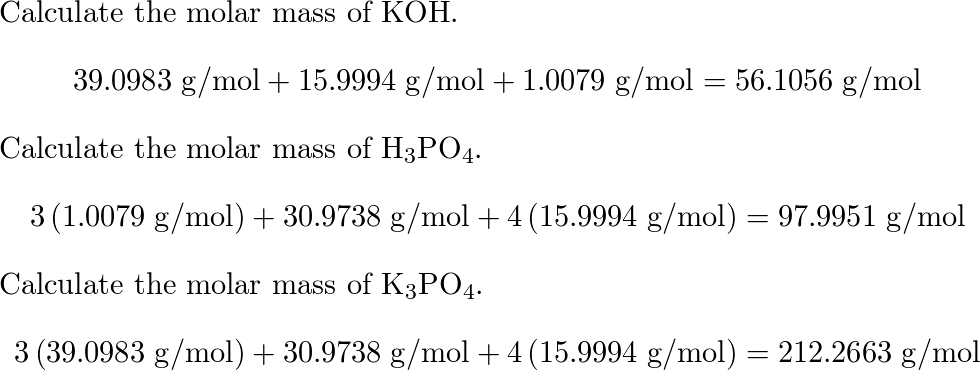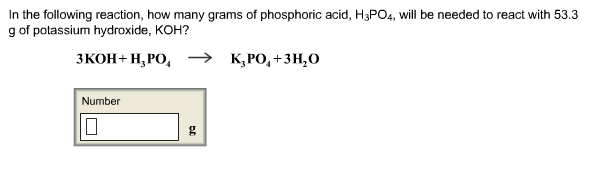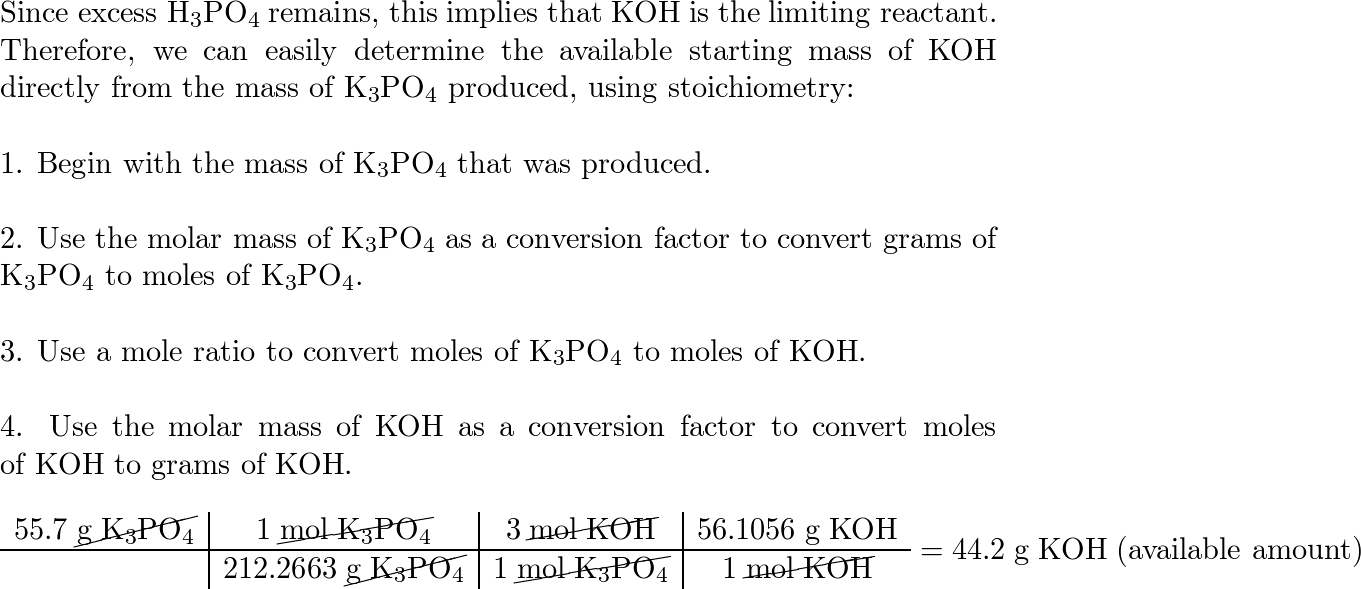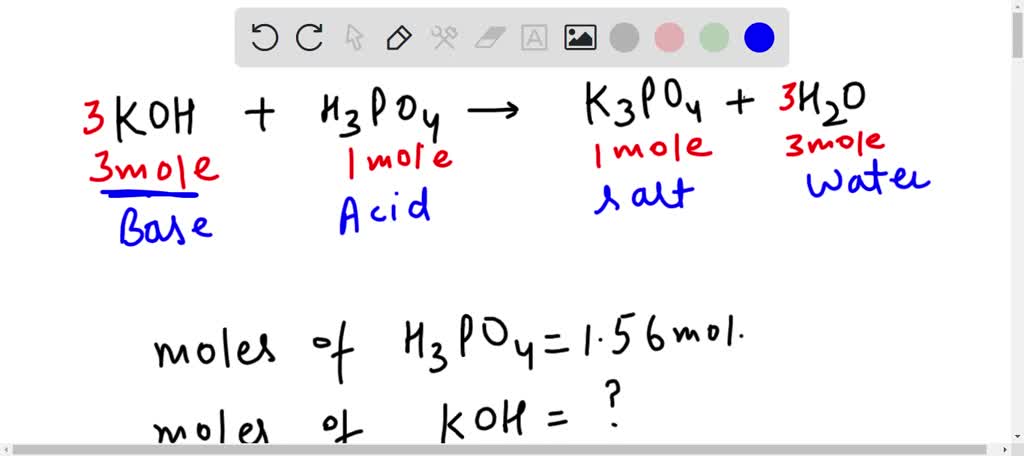
SOLVED: How many moles of potassium hydroxide (KOH) are needed to completely neutralize 1.56 mol phosphoric acid (H3PO4)?

Outline of AC preparation from RH. In figure, KOH, ZnCl2, H3PO4, and N... | Download Scientific Diagram

OneClass: 2.For the reaction of phosphorous acid (H3PO3) and potassium hydroxide (KOH), write (a) the...
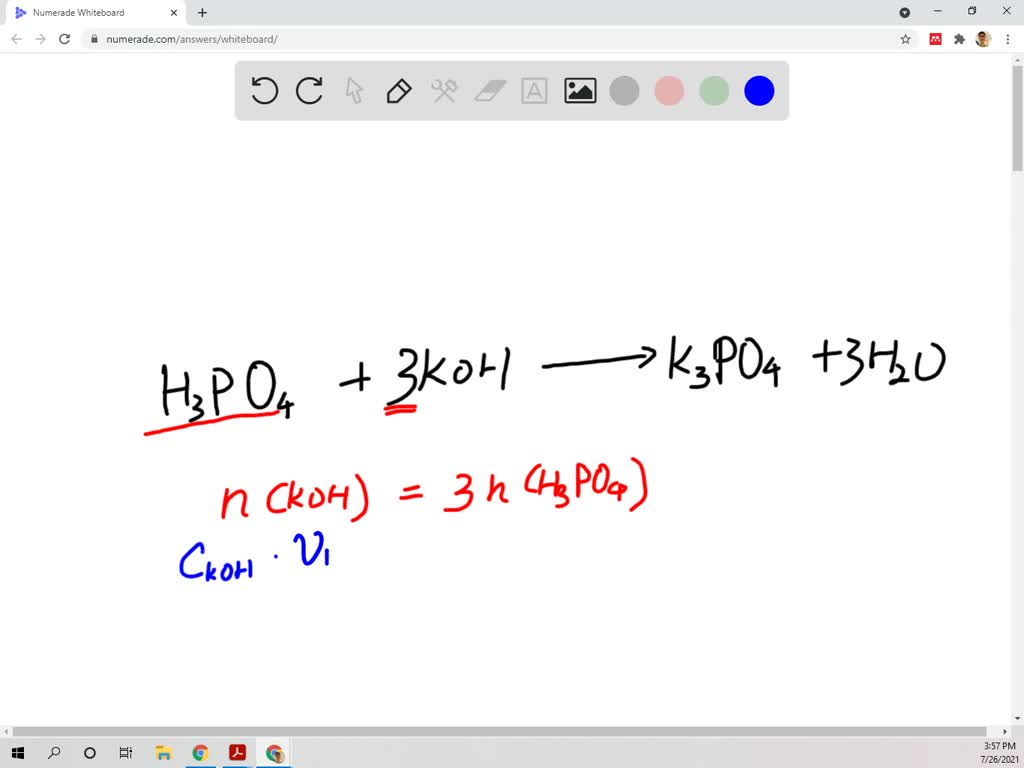
SOLVED: A 0.0700 L sample of phosphoric acid (H3PO4) solution with an unknown concentration reacts with 0.200 L of 0.300 M potassium hydroxide ( KOH) solution. What is the concentration of the phosphoric
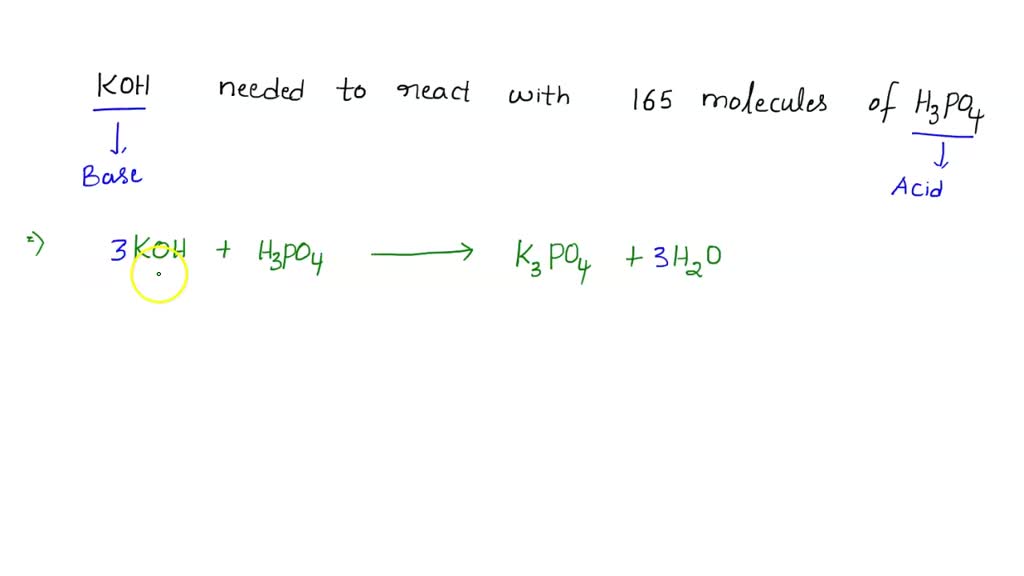
SOLVED: 'How many units of potassium hydroxide are needed to react with 165 molecules of phosphoric acid? BKOHq) HzPO4aq) KzPO4aq) 3HzQ()'

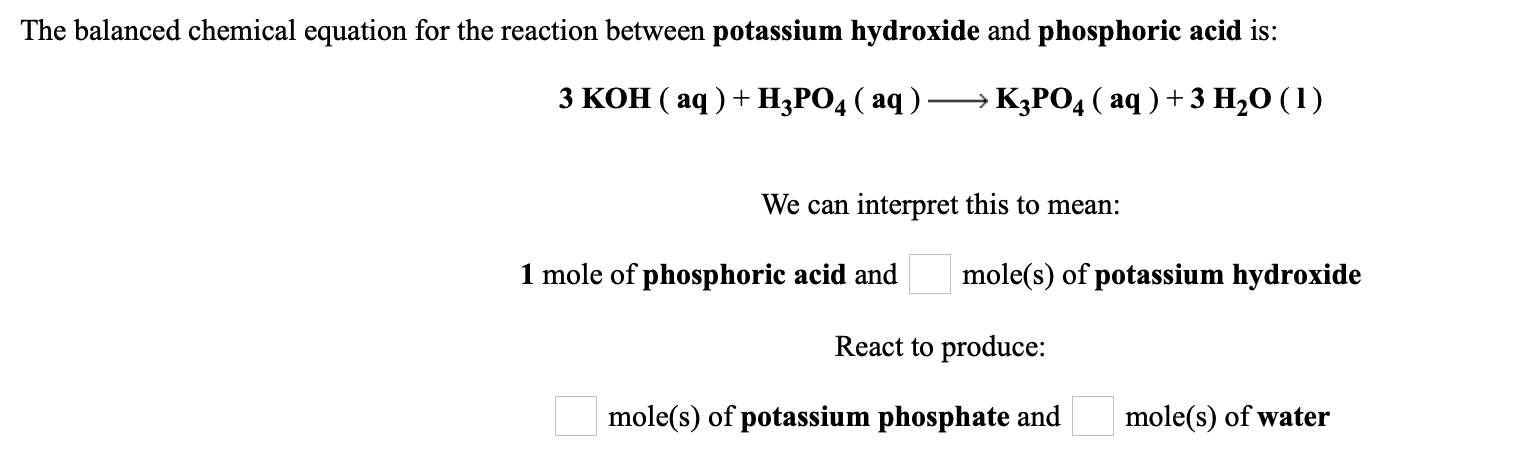
SOLVED: 28)| a) When potassium hydroxide reacts With phosphoric acid potassium phosphate and water are produced The balanced equation for this reaction is 3KOH(aq) + H;PO (aq) K;POA (aq) + 3H20() If
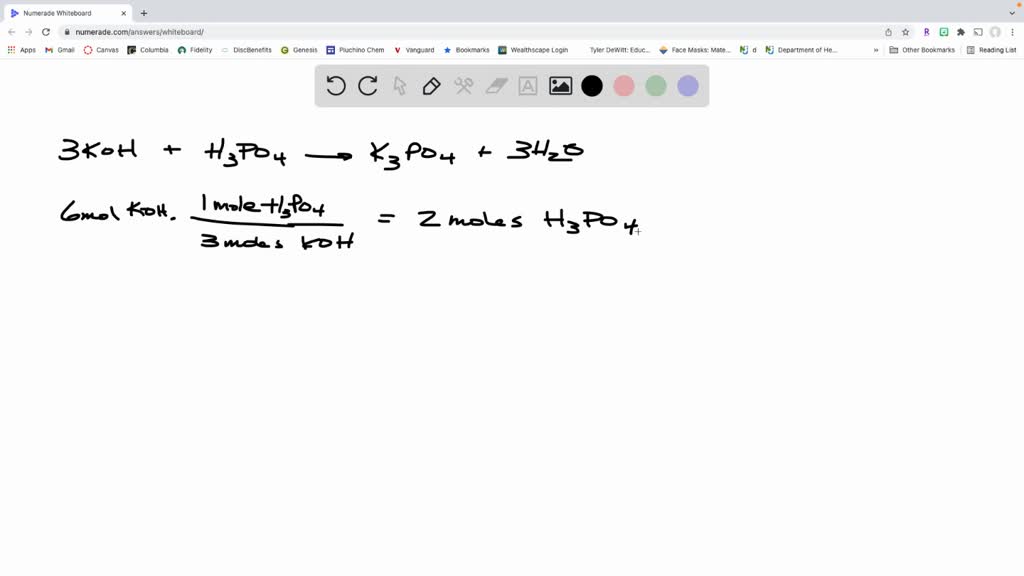
SOLVED: When potassium hydroxide reacts with phosphoric acid, potassium phosphate and water are produced. The balanced equation for this reaction is: 3KOH(aq) + H3PO4 (aq) -> K3PO4(aq) + 3H2O(l) If 6 moles
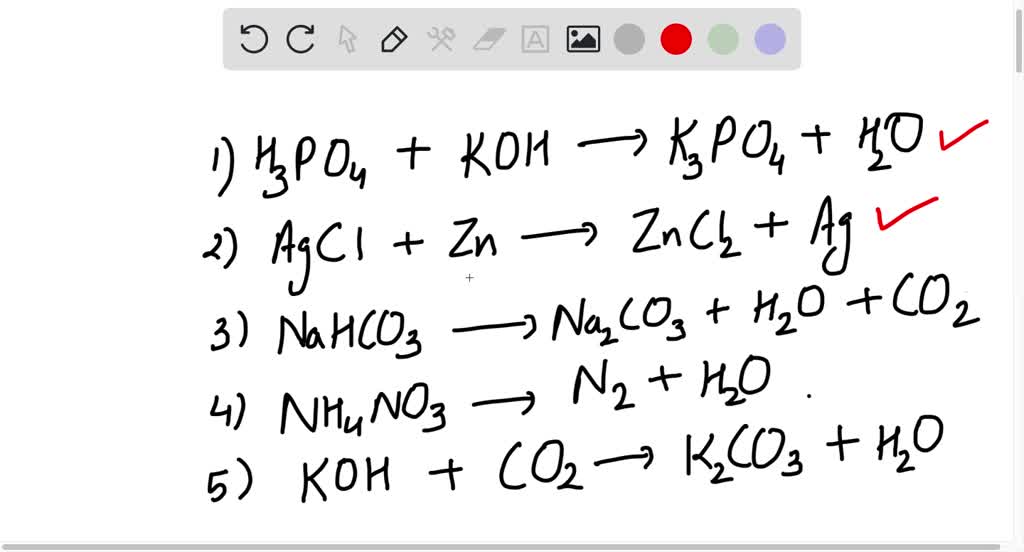
SOLVED: Write an unbalanced equation to represent each of the following reactions: (a) Potassium hydroxide and phosphoric acid react to form potassium phosphate and water. (b) Zinc and silver chloride react to