
The equivalent conductances of sodium choride, hydrochloric acid and sodium acetate at infinite dilution are 126.45, 426.16 and 91.0 ohm^-1 cm^2 eq^-1 , respectively, at 25^∘C . Calculate the equivalent conductance of

20 ml of 0.1 M acetic acid is mixed with 10 ml of sodium acetate. pKa of CH3COOH is 4.8 , then the concentration of sodium acetate, if pH of the mixture is 4.8 is:

SOLVED: buffer solution is made from acetic acid, HCzH;Oz, and sodium acetate, NaCzH;Oz: Suppose small amount of hydrochloric What is the net ionic equation for the reaction that occurs when acid is
✓ Solved: A quantity of 0.15 M hydrochloric acid is added to a solution containing 0.10 mol of sodium...

OneClass: If we have a 25.00ml solution containing .038M sodium acetate, NaCH3COO, how much .098M HCl...

SOLVED: What reaction occurs as hydrochloric acid solution is added to a solution containing equal concentrations of acetic acid and sodium acetate? CH; COOH + Ht Lo CH;COOH2 b CH;COO-+H 7 CH;COOH

Titration of CH3COONa with HCl and pKa determination from half equivalence point - Chemistry Stack Exchange

State and exlain Kohlarausch.s law of independent migration of ions . The equivalent conductance of infinite dilution (A(0)) for sodium acetate , sodium chloride and hydrochloric acid are 78, 109 and 384 ohm^(-1)cm^(2) g. Calculate the A(0) of acetic acid .
When a small amount of HCL is added to a buffer solution of acetic acid and sodium acetate what happen?
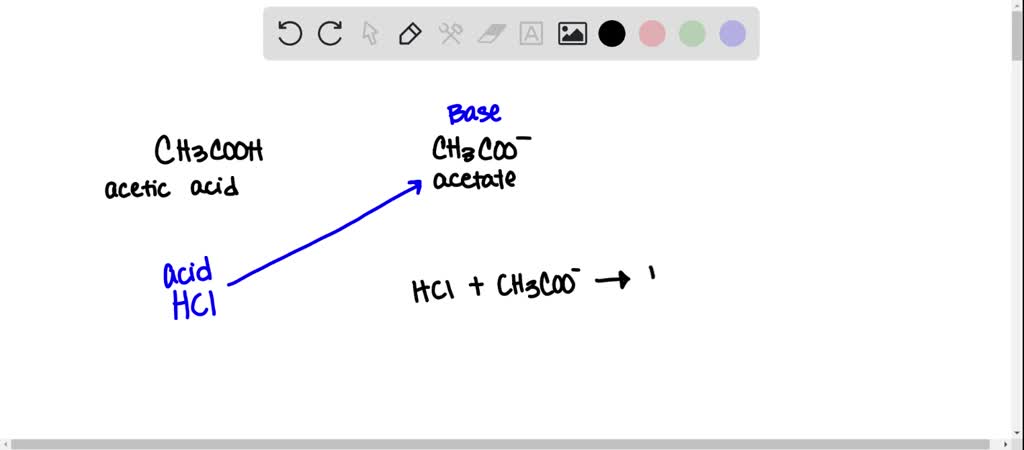
SOLVED: Q: When hydrochloric acid solution is added to a solution containing equal concentrations of acetic acid and sodium acetate, how is the pH maintained? a. The acid of the buffer reacts

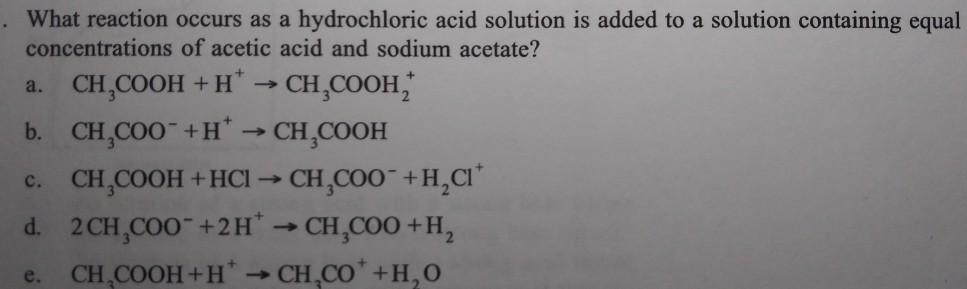






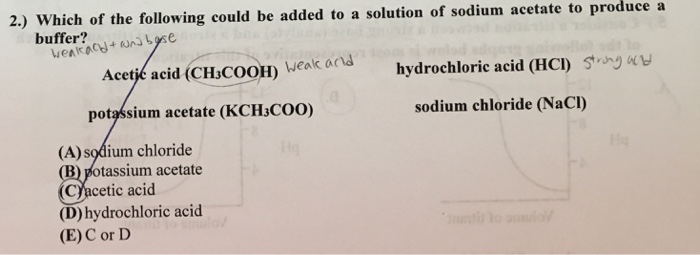




![Sodium Acetate [CH3COONa] Molecular Weight Calculation - Laboratory Notes Sodium Acetate [CH3COONa] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2021/11/sodium-acetate-molecular-weight-calculation.jpg)
