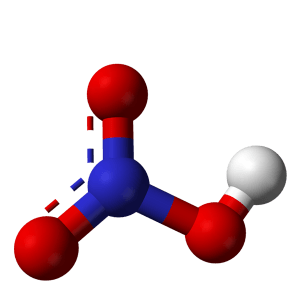
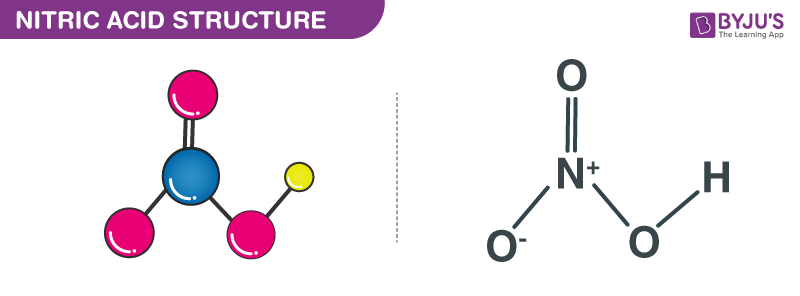
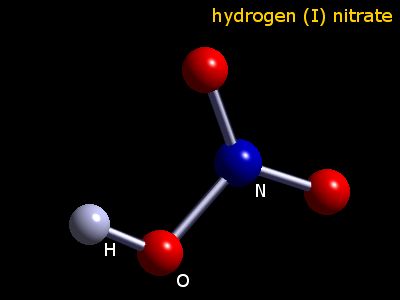
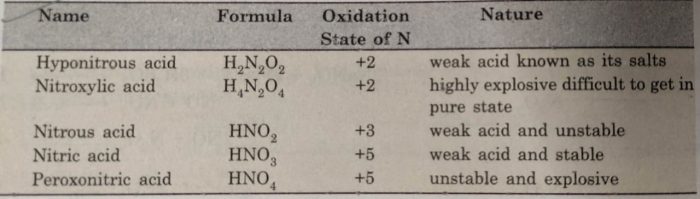
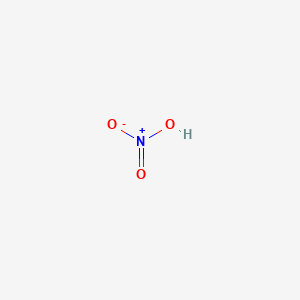
I made some lead II nitrate by dissolving lead metal in nitric acid and recrystallised the solid salt. This video demonstrates the thermal decomposition of lead ii nitrate. I also did a
Ozone-nitric acid tracer-tracer correlation. Solid red line represents... | Download Scientific Diagram

Liquid–Liquid Equilibrium in the Nitric Acid + Calcium Nitrate + Water + Tri-n-octylamine System | Journal of Chemical & Engineering Data

Structure of Nitric Acid in Vapor & Solid Phases || CH#4 || P-Block Elements || XII - Chemistry - YouTube
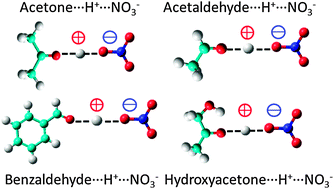
Proton transfer reactions between nitric acid and acetone, hydroxyacetone, acetaldehyde and benzaldehyde in the solid phase - Physical Chemistry Chemical Physics (RSC Publishing)