Teva Secures European Approval of Trisenox for First Line Treatment of Low to Intermediate Risk Acute Promyelocytic Leukemia » FINCHANNEL

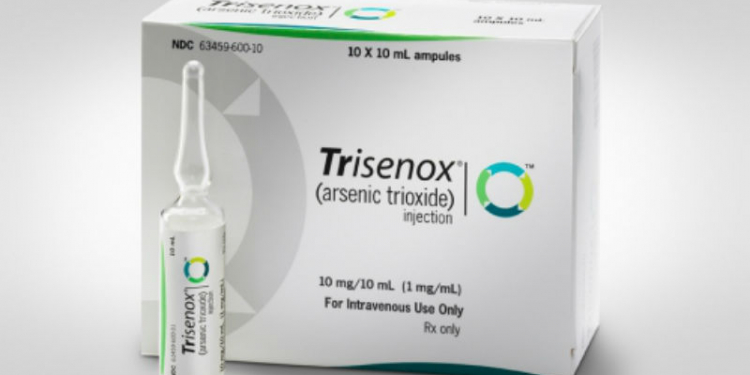
Arsenio 2mg/ML c/ 10 amp-06ml IV - Trisenox Teva - ÁgilMed - Medicamentos Especiais e Nutrição Clínica
Teva Receives FDA Priority Review for First Line Use of TRISENOX® (arsenic trioxide) in Patients with Low to Intermediate Risk Acute Promyelocytic Leukemia (APL) | media
Fresenius Kabi Introduces Leukemia Drug Arsenic Trioxide Injection in 10 mg per 10 mL vial | Business Wire
Direct Healthcare Professional Communication TRISENOX (arsenic trioxide) – Risk of medication errors due to the introduction o
Direct Healthcare Professional Communication TRISENOX (arsenic trioxide) – Risk of medication errors due to the introduction o
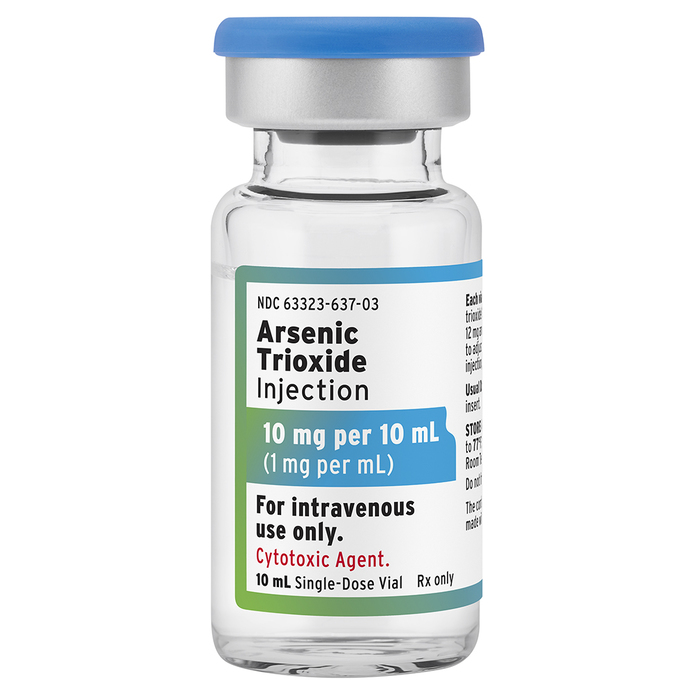
Fresenius Kabi Introduces Leukemia Drug Arsenic Trioxide Injection in 10 mg per 10 mL vial - Fresenius Kabi USA
1 TRISENOX (arsenic trioxide) – Risk of medication errors due to the introduction of 2 mg/ml concentration: New concentration:

Global Trisenox (arsenic trioxide) Market Growth 2018-2023 | Market research, Marketing, Stock market

EU approval of Teva's Trisenox® for first line treatment of low to intermediate risk APL - Hospital Pharmacy EuropeHospital Pharmacy Europe

How a drug for few patients was turned into $81 million in sales | Pharma's Windfall | The Seattle Times


/teva_pag.png?sfvrsn=bb09d0ac_2)