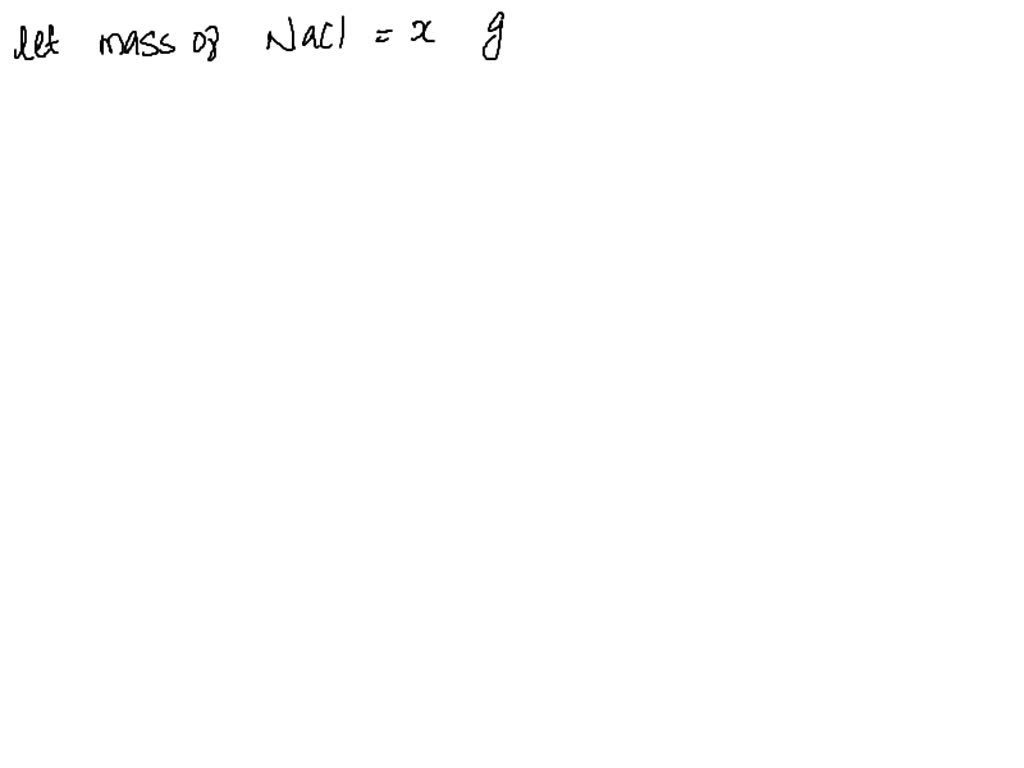
SOLVED: A 0.1824-g sample that contains both NaCl and NaBr is titrated with 0.1052 M AgNO3, requiring 26.48 mL. Calculate the percentages of NaCl and NaBr in the sample. NaCl = 58.442 NaBr = 102.89

Effect of NaCl and KCl on the Solubility of Amino Acids in Aqueous Solutions at 298.2 K: Measurements and Modeling | Industrial & Engineering Chemistry Research

Comparison of the contact angle variation between NaCl and KCl solution... | Download Scientific Diagram

Raw and cooked noodles prepared with NaCl and LMC-salt. (a) Raw noodle... | Download Scientific Diagram
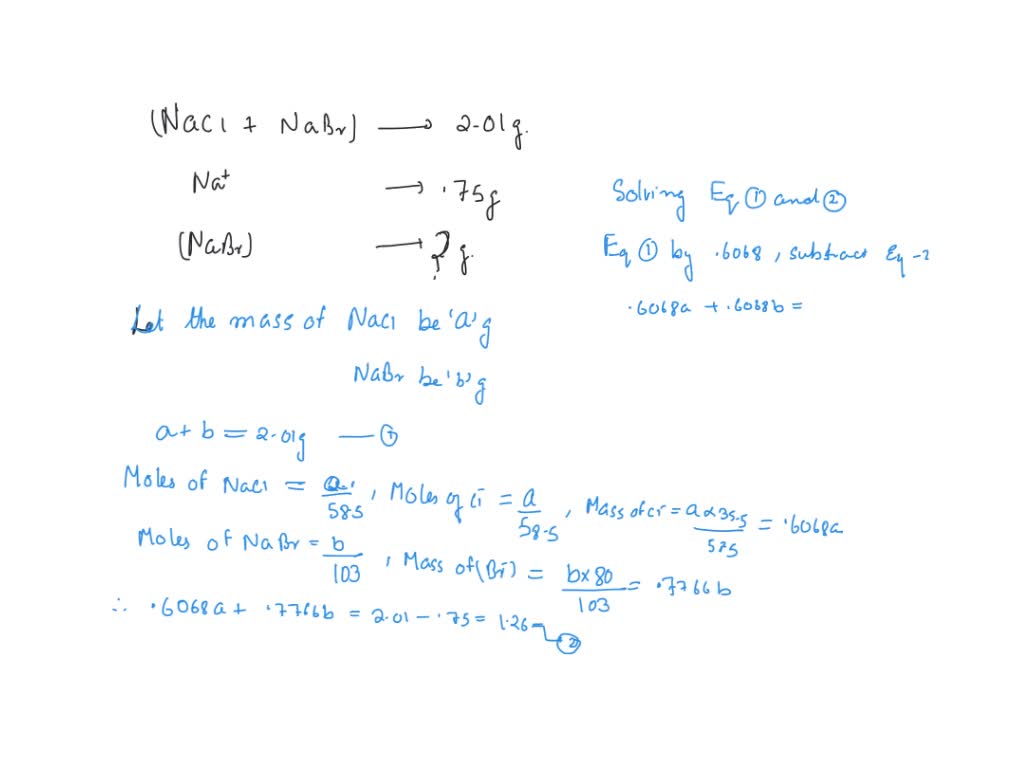
SOLVED: A mixture of NaCl and NaBr has a mass of 2.01 g and is found to contain .75 grams of Na. What is the mass of NaBr in the mixture?

Dissolution of calcite in 3 m NaCl and 1 m CaCl 2-2 m NaCl solutions at... | Download Scientific Diagram

Germination percentage and germination index of E. sativa seeds under... | Download Scientific Diagram

Total oil recovery from the sandstone core after flooding with brine... | Download Scientific Diagram

SOLVED: Suppose you have a mixture of sodium chloride, NaCl, and carbon, C. Explain how you can use water to separate the two substances.
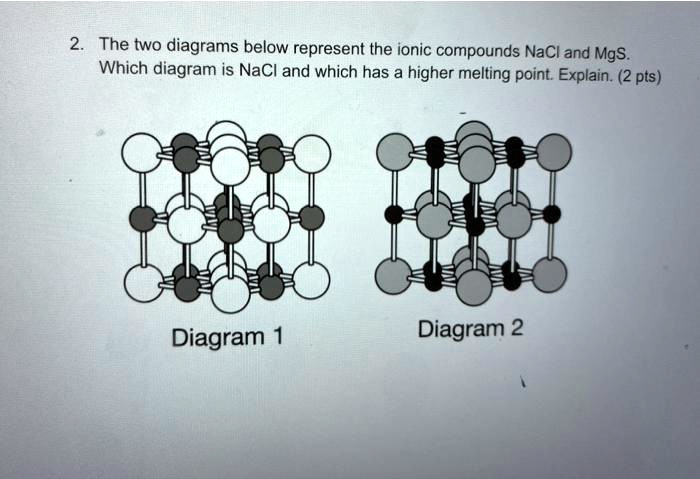
SOLVED: The tWO diagrams below represent the ionic compounds NaCl and Mgs Which diagram is NaCI and which has a higher melting point: Explain; 2 pts) Diagram 1 Diagram 2

A SEM picture of porous HA after the removal of NaCl and PVA as pore... | Download Scientific Diagram

SOLVED: A 0.8870 g sample of a mixture of NaCl and KCl is dissolved in water, and the solution is then treated with an excess of AgNO3 to yield 1.782 g of
![SOLVED: 10.0 g of NaCl and 100.0 g of Select ] HzO 50.0 mL of ethanol, CzHsOH and Water is the solute and ethan 10.0 mL of HzO 0.20 Lof 02 and SOLVED: 10.0 g of NaCl and 100.0 g of Select ] HzO 50.0 mL of ethanol, CzHsOH and Water is the solute and ethan 10.0 mL of HzO 0.20 Lof 02 and](https://cdn.numerade.com/ask_images/4fa281ca316d40fda76e19c1f8aca2f8.jpg)
SOLVED: 10.0 g of NaCl and 100.0 g of Select ] HzO 50.0 mL of ethanol, CzHsOH and Water is the solute and ethan 10.0 mL of HzO 0.20 Lof 02 and

CFMEU hopes preferencing Labor last will send message to State Government over New Acland near Oakey, west of Toowoomba | The Chronicle

Amazon.com: HiMedia M082A-500G MacConkey Agar Without CV, NaCl and with 0.5% Bile Salts, 500 g : Industrial & Scientific

SOLVED: If you start with mixture of NaCl and KCIO3 that weighs 3.00 g and the mass of the sample decreases by 0.75 g after heating, what is the best explanation for
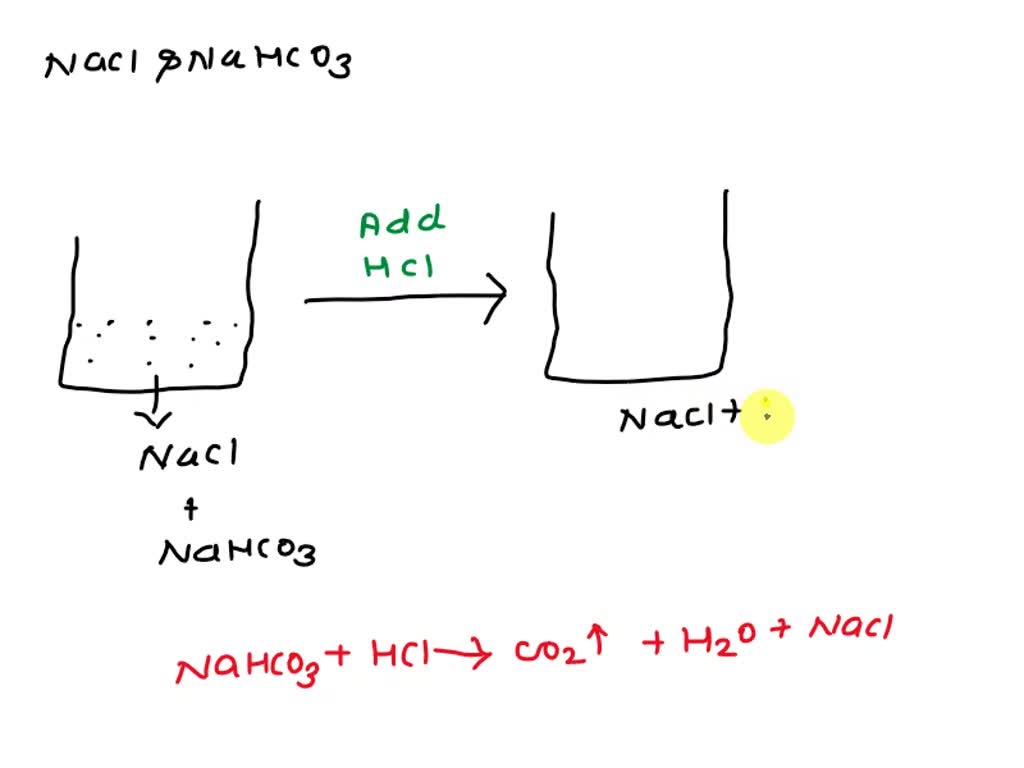
SOLVED: What is the best way for separation of a mixture NaCl and NaHCO3 in 200 mL water? HCl is available and we need to separate NaCl and water in two different

Manipulation of NPs over a Au film in NaCl and SDS solution a AuNP with... | Download Scientific Diagram
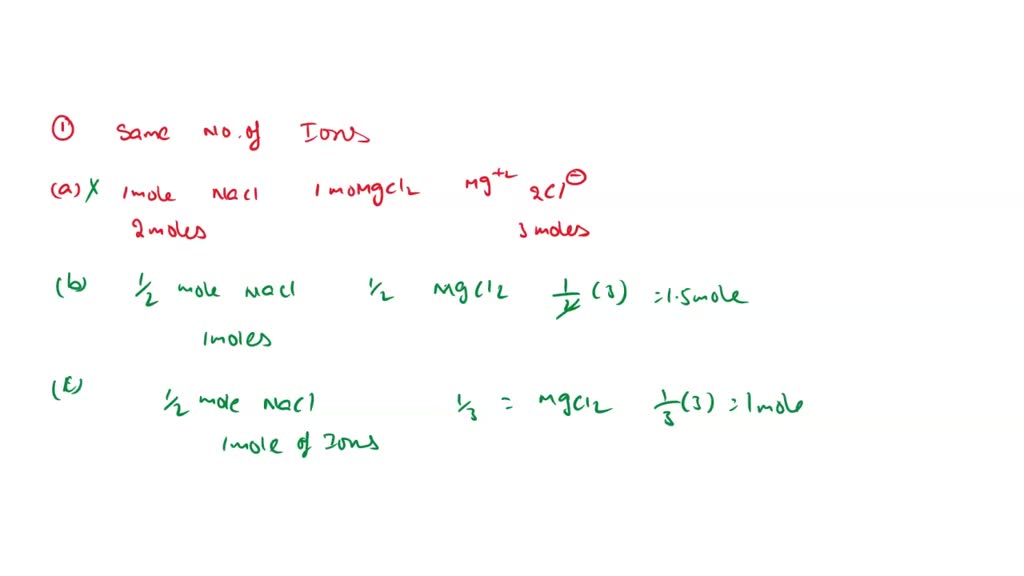
SOLVED: Whi(a) I mole of NaCl and I mole of MgCl2(b) 1/2 mole of NaCl and 1/2 mole of MgCl2(c) 1/2 mole of NaCl and 1/3 mole of MgCl2(d) 1/3 mole of

rsa1-1 plants are hypersensitive to NaCl, and RSA1 is involved in Na+... | Download Scientific Diagram
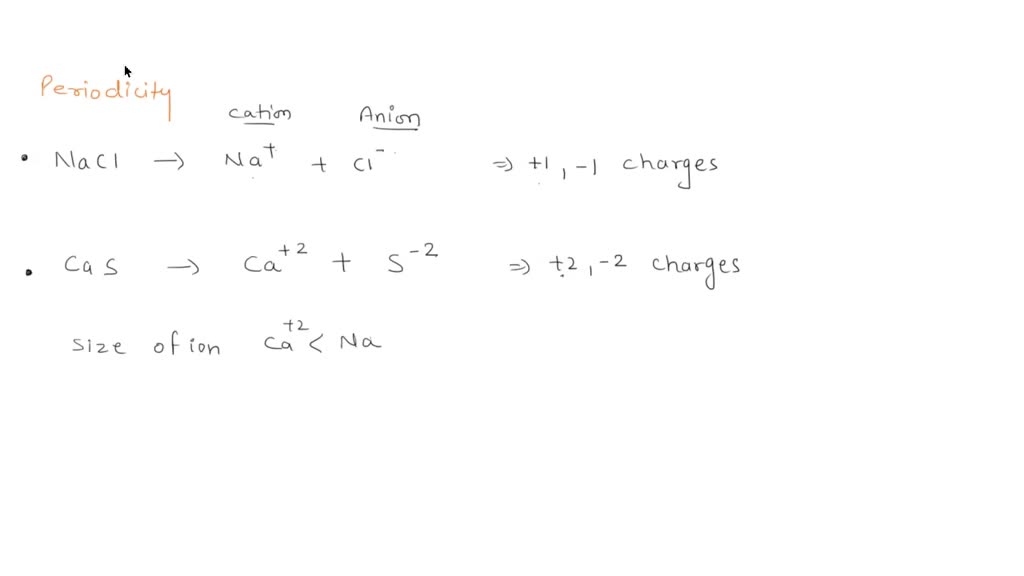
SOLVED: between NaCl and CaS, which do you think has a higher melting point? use the concepts of periodic trends and ionic bonding to justify your answer

NaCl and KCl mediate log increase in AAV vector particles and infectious titers in a specific/timely manner with the HSV platform - ScienceDirect
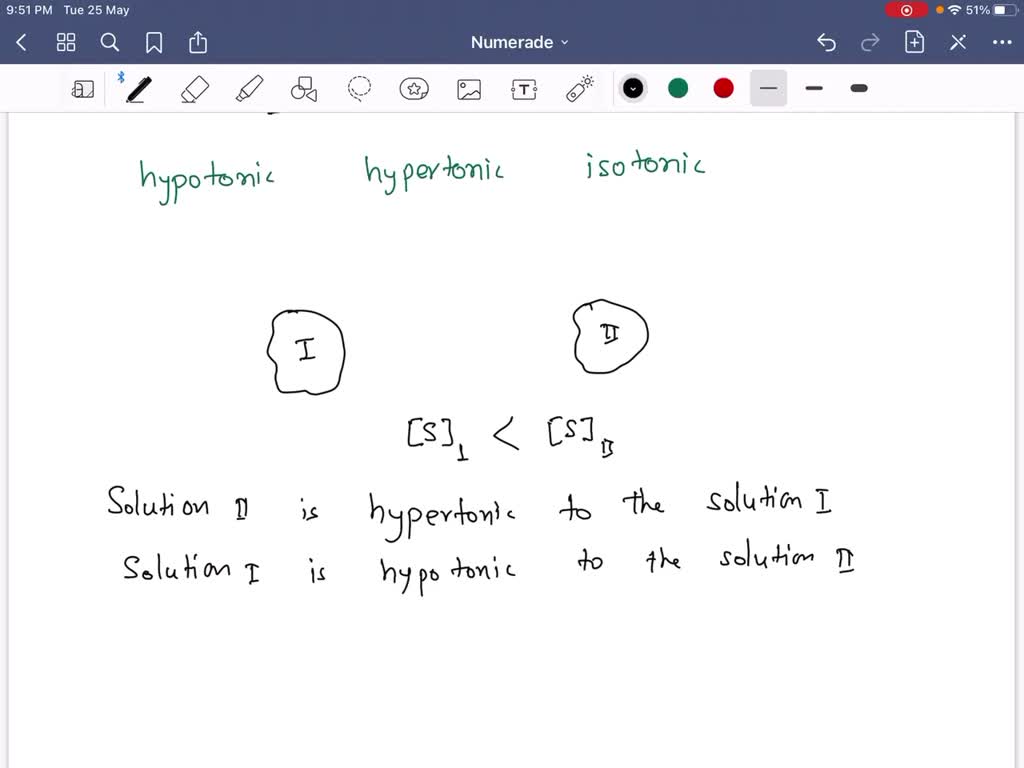
SOLVED: A cell is placed in a beaker containing a solution of 40% NaCl and 60% water. After a few minutes, you notice that the cytoplasm of the cell is shrinking in


