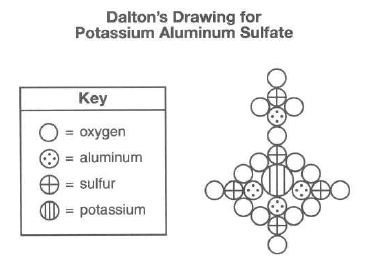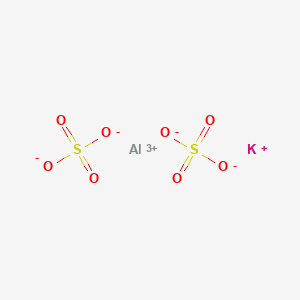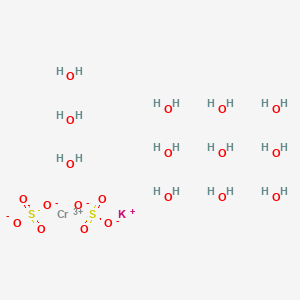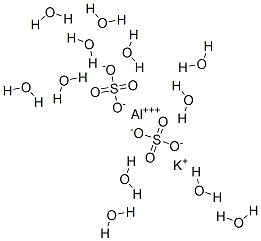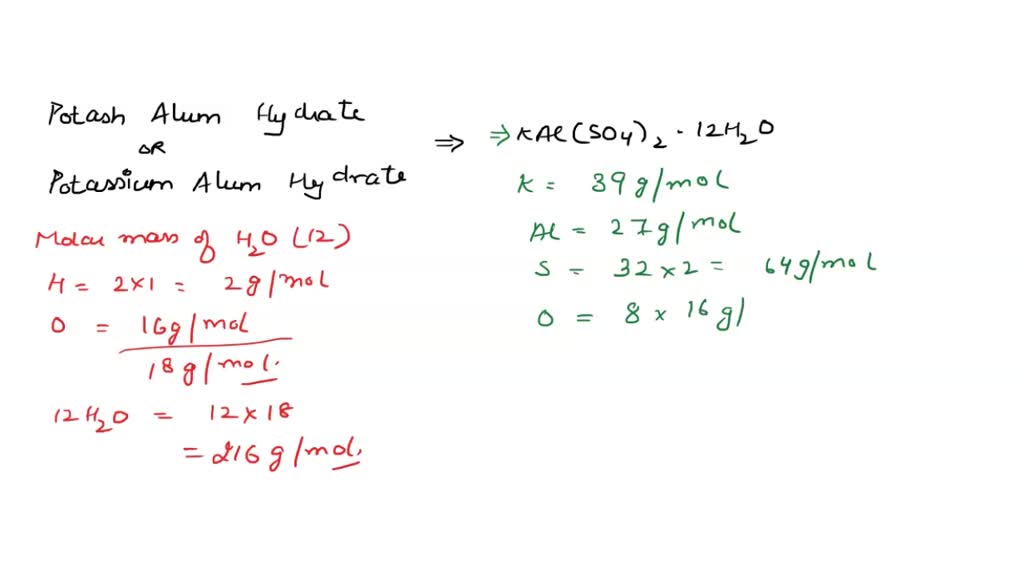
SOLVED: What is the mass percent of water in the Potassium Alum Hydrate: KAl (SO4)2·12H2O? A) 8.33% B) 91.67% C) 21.62% D) 54.43% E) 45.57%

Question Video: Calculating the Percentage by Mass of Water in Alum Given Its Chemical Formula | Nagwa
![Potassium Alum [KAl(SO4)2∙12H2O] solid catalyst for effective and selective methoxylation production of alpha-pinene ether products - ScienceDirect Potassium Alum [KAl(SO4)2∙12H2O] solid catalyst for effective and selective methoxylation production of alpha-pinene ether products - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S2405844021001638-sc1.jpg)
Potassium Alum [KAl(SO4)2∙12H2O] solid catalyst for effective and selective methoxylation production of alpha-pinene ether products - ScienceDirect
Types of the coordination polyhedra of K + in K(Al,Cr)(SO 4 ) 2 ⋅12H 2... | Download Scientific Diagram
Potassium Alum [KAl(SO4)2∙12H2O] solid catalyst for effective and selective methoxylation production of alpha-pinene

A) on a molecular scale, describe how a crystal of alum differs from a crystal of potassium aluminum - Brainly.com

Polarized Raman Spectra of NH𝟒Al(SO𝟒)𝟐⋅𝟏𝟐H𝟐O – topic of research paper in Nano-technology. Download scholarly article PDF and read for free on CyberLeninka open science hub.
Crystal Chemistry and High-Temperature Behaviour of Ammonium Phases NH4MgCl36H2O and (NH4)2Fe3+Cl5H2O from the Burned Dumps of t
Thermal behaviour of alum-(K) KAl(SO4)2·12H2O from in situ laboratory high-temperature powder X-ray diffraction data: thermal e

Investigations on KAl(SO4)2∙12H2O: A Candidate α-Alum Material for Energy Storage Applications | SpringerLink
A study of the relaxation mechanism of a KAl(SO4)2·12H2O single crystal by observation of its 39K and 27Al spin–lattice relax