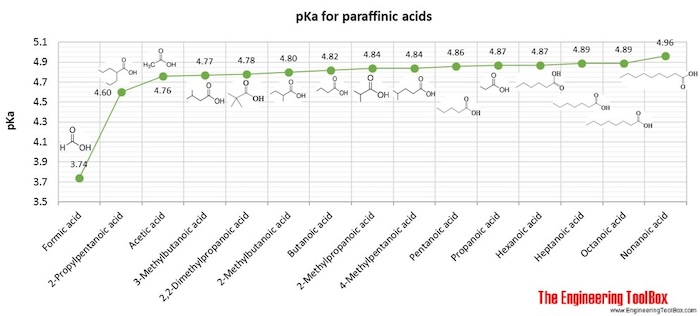
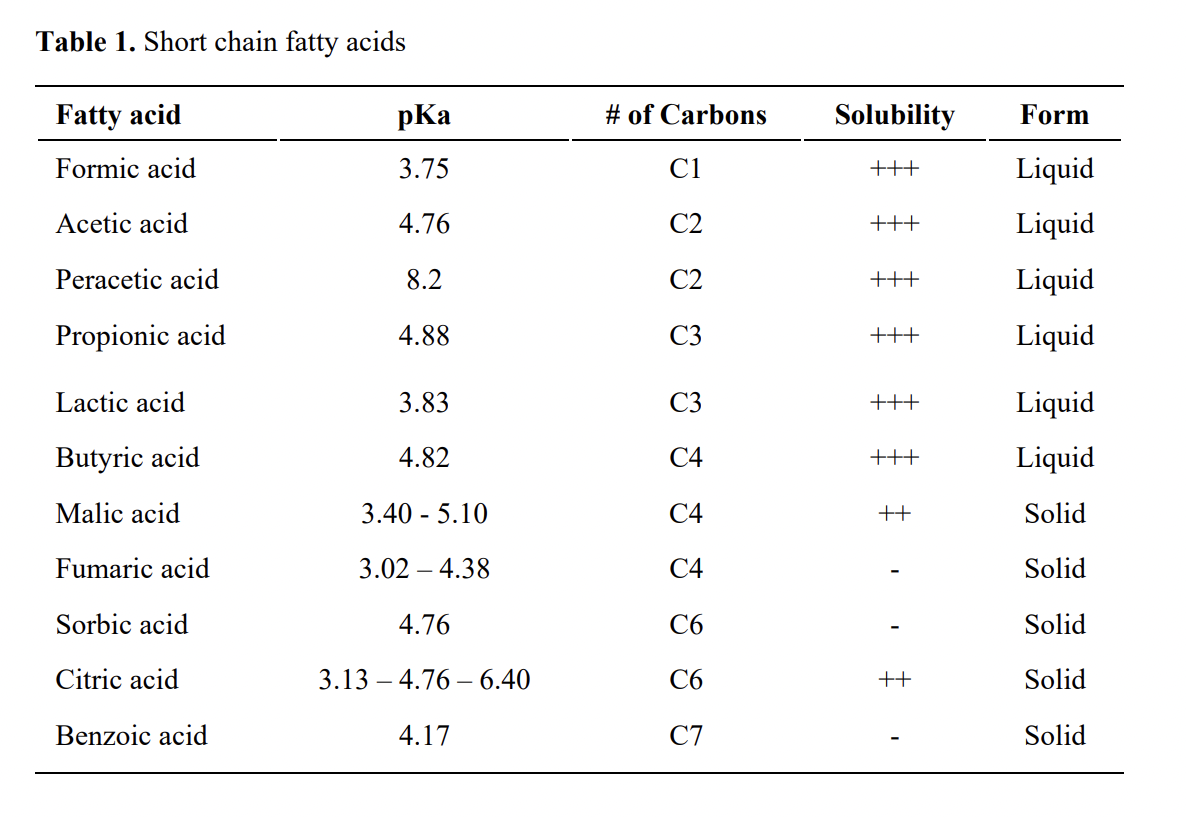
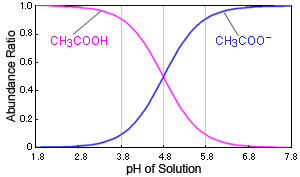
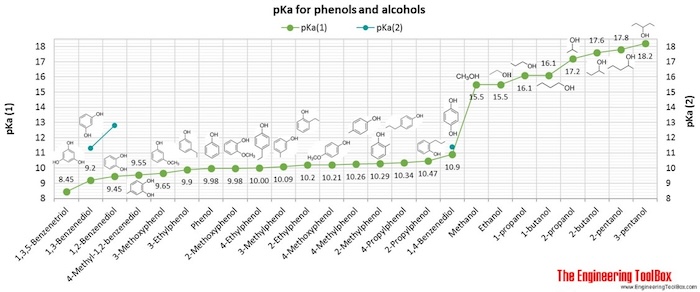
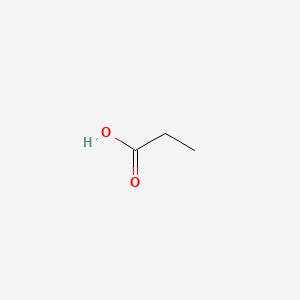
Why is the pka of vanillin lower than salicylic aldehyde, which is lower than the pka of a carboxylic acid (ex: propionic acid)? : r/OrganicChemistry

List of organic acids and their properties, list of acids and their... | Download Scientific Diagram

SOLVED: pKa of propionic acid is 4.87. What is the pH of a buffer that is 0.24 propionic acid ( H3C- CHs-COOH) and 0.29 M sodium propanate (H3C-CH2-COONa).

Empirical Conversion of pKa Values between Different Solvents and Interpretation of the Parameters: Application to Water, Acetonitrile, Dimethyl Sulfoxide, and Methanol | ACS Omega
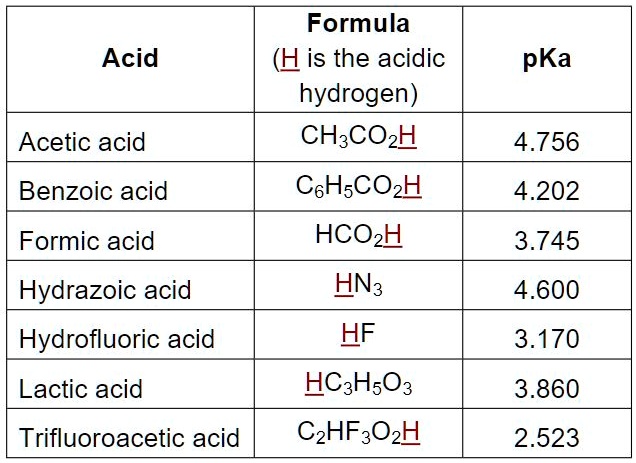
SOLVED: Formula Acid (His the acidic hydrogen) CH:COzH pKa Acetic acid 4.756 Benzoic acid CsHsCOzH HCOzH HN3 HF 4.202 Formic acid 3.745 Hydrazoic acid Hydrofluoric acid Lactic acid 4.600 3.170 HCHsO3 3.860 Trifluoroacetic acid CzHF3OzH 2.523
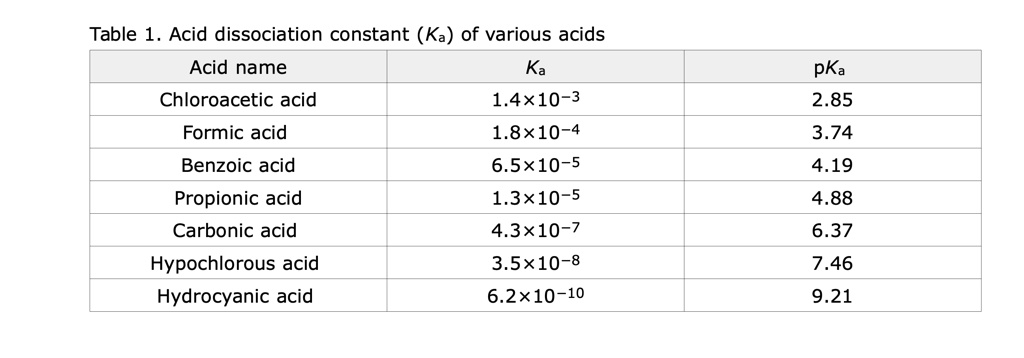
SOLVED: Table Acid dissociation constant (Ka) of various acids Acid name Ka Chloroacetic acid 1.4x10-3 Formic acid 1.8*10-4 Benzoic acid 6.5x10-5 Propionic acid 1.3x10-5 Carbonic acid 4.3*10-7 Hypochlorous acid 3.5x10-8 Hydrocyanic acid
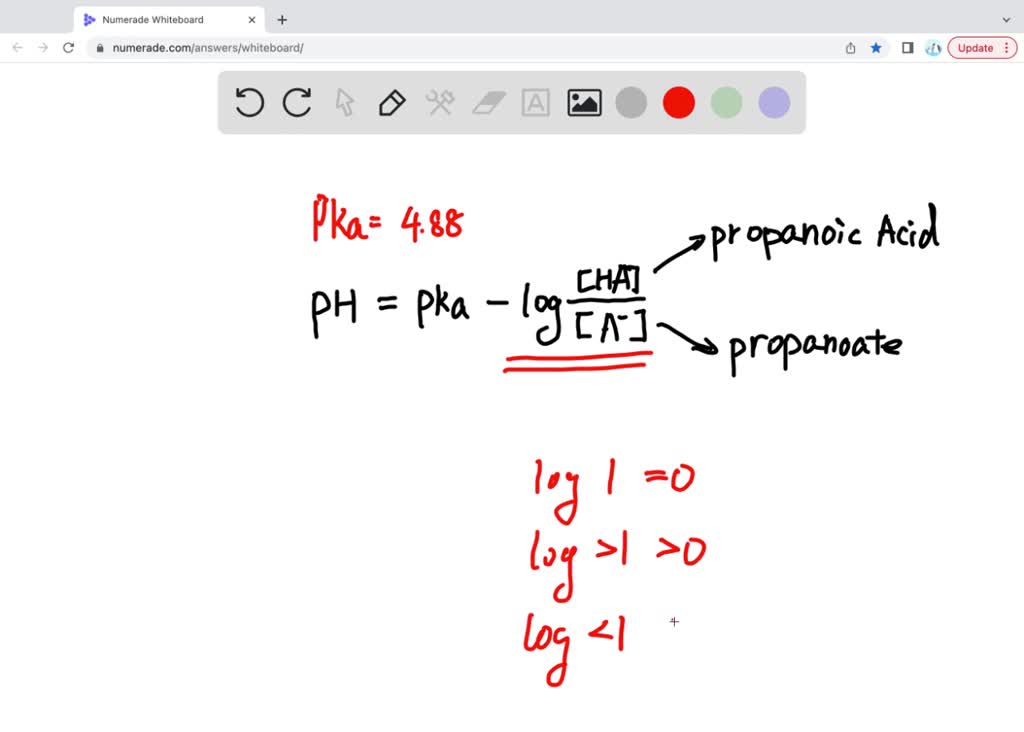
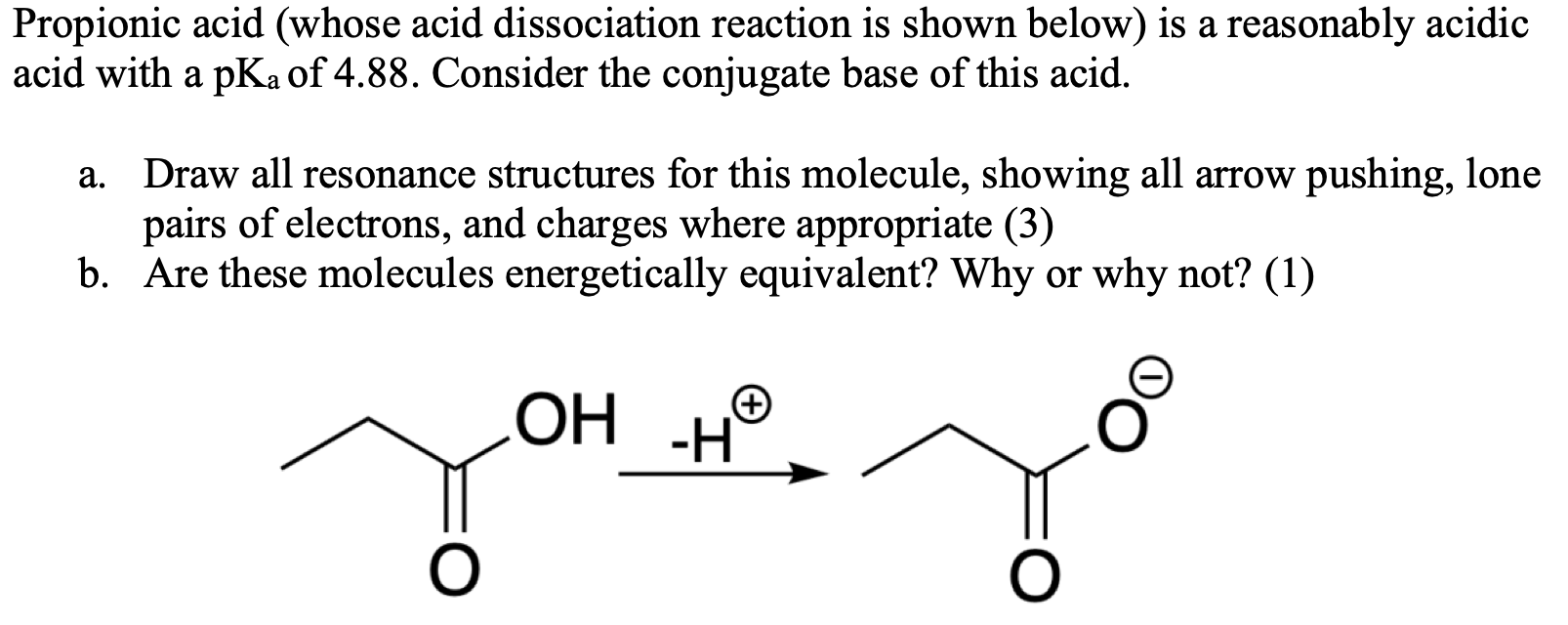
SOLVED: 'The pKa of propanoic acid is 4.88. Describe what the relative concentrations of propanoic acid (the protonated form of this acid) and propanoate (its conjugate base) will be (1) at; (2)

Match each of the following pKa values (3.2, 4.9, and 0.2) to the appropriate carboxylic acid: (a) CH3CH2COOH; (b) CF3COOH; (c) ICH2COOH. | Homework.Study.com